Stability Chambers
1. Stability Studies and Regulatory Framework
Stability studies are conducted to determine how the quality of a pharmaceutical product changes over time when exposed to environmental conditions such as temperature, humidity, and light. The purpose of these studies is to establish product shelf life, recommended storage conditions, and packaging suitability throughout the product lifecycle.
Regulatory authorities require stability data to demonstrate that a drug product maintains its identity, strength, quality, and purity during the entire labeled storage period. Stability programs therefore involve storing representative product samples under defined environmental conditions and performing periodic analytical testing to evaluate potential changes in critical quality attributes. These attributes may include potency, degradation products, dissolution characteristics, physical appearance, moisture content, and microbiological quality.
Stability study design and environmental conditions are defined in international regulatory guidance documents. Key guidance documents include:
- ICH Q1A(R2) Stability Testing of New Drug Substances and Products
- ICH Q1B Photostability Testing of New Drug Substances and Products
- ICH Q1D Bracketing and Matrixing Designs for Stability Testing
These guidelines define the environmental conditions used for long-term, intermediate, and accelerated stability testing and form the basis for regulatory stability programs in pharmaceutical development and manufacturing.
2. Overview of Stability Chambers
Stability chambers are controlled environmental systems used to maintain the temperature and relative humidity conditions required for pharmaceutical stability studies. These chambers provide tightly regulated environments in which product samples are stored for extended periods while stability testing is performed according to approved stability study protocols.
Unlike incubators or warm rooms that primarily control temperature, stability chambers regulate multiple environmental parameters simultaneously. Environmental control systems typically maintain temperature, relative humidity, and airflow distribution to ensure that conditions remain consistent throughout the chamber volume. Typical operating conditions used in pharmaceutical stability chambers include:
- 25 °C / 60 % RH for long-term stability testing
- 30 °C / 65 % RH for intermediate stability conditions
- 40 °C / 75 % RH for accelerated stability testing
Because stability studies may continue for many months or years, stability chambers must maintain stable environmental conditions with high reliability and minimal variation. Continuous environmental monitoring and alarm systems are therefore essential to ensure that defined stability conditions are maintained throughout the study duration.
3. Types of Stability Chambers
Stability chambers are available in several configurations depending on storage capacity, environmental control capability, and intended application.
3.1 Reach-In Stability Chambers
Reach-in stability chambers resemble laboratory refrigerators or incubators and provide controlled environmental conditions within a compact cabinet enclosure. These chambers typically include multiple adjustable shelves and are used for smaller stability studies, development work, or storage of limited quantities of samples. Reach-in chambers offer:
- compact footprint
- precise environmental control
- flexibility for laboratory environments
They are frequently used during early product development or for supplemental stability studies. The diagram below shows a typical reach-in pharmaceutical stability chamber used for controlled temperature and humidity storage of stability samples. These chambers provide regulated environmental conditions and include integrated temperature and humidity control systems, circulation fans, and monitoring sensors to maintain uniform environmental conditions across storage shelves.

3.2 Walk-In Stability Chambers
Walk-in chambers provide large controlled environments designed to accommodate extensive stability programs involving many product batches or packaging configurations. These chambers are constructed as insulated rooms with integrated environmental control systems that regulate temperature and humidity throughout the entire chamber volume.
The diagram below illustrates the environmental control architecture used in stability chambers. Environmental control systems regulate temperature and relative humidity through coordinated operation of refrigeration units, heaters, humidification systems, dehumidification components, and air circulation fans to maintain stable chamber conditions.

Walk-in chambers are commonly used in pharmaceutical manufacturing facilities where long-term stability programs require storage of large numbers of stability samples. Typical features include:
- environmental control HVAC systems
- high-capacity humidity generation systems
- circulation fans for uniform air distribution
- automated monitoring and alarm systems
3.3 Photostability Chambers
Photostability chambers are specialized systems used to evaluate the effect of light exposure on pharmaceutical products. These chambers combine controlled temperature conditions with defined illumination levels in accordance with regulatory guidance such as ICH Q1B. Lighting systems within photostability chambers deliver controlled ultraviolet and visible light exposure to simulate conditions that may cause photodegradation of drug products and packaging materials.
The image below shows a typical pharmaceutical photostability chamber used in stability studies. These chambers expose samples to controlled light sources while maintaining stable environmental conditions to support photostability testing performed according to ICH Q1B requirements.

4. Environmental Control Architecture
The environmental control architecture of a stability chamber integrates multiple systems that regulate temperature and relative humidity within the controlled environment. These systems operate together to maintain stable environmental conditions required for pharmaceutical stability studies. Typical environmental control components include:
- refrigeration systems for temperature reduction
- electric heaters for temperature control
- humidification systems to generate controlled moisture levels
- dehumidification systems to remove excess moisture
- air circulation fans to distribute conditioned air uniformly
The diagram below illustrates the environmental control architecture used in a typical stability chamber, showing how heating, cooling, humidification, and dehumidification components interact with the air circulation system and environmental sensors.
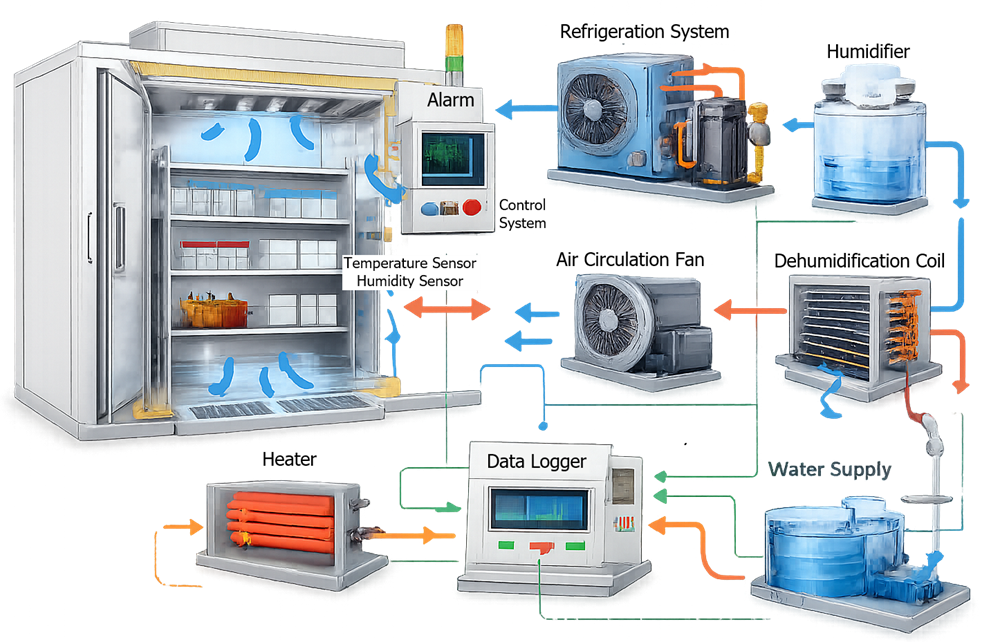
Temperature and humidity sensors continuously monitor chamber conditions. Signals from these sensors are transmitted to the control system, which adjusts heating, cooling, humidification, and dehumidification functions to maintain the defined environmental setpoints.
4.1 Temperature Control
Temperature regulation is achieved through coordinated operation of refrigeration compressors and heating elements. The control system modulates these components to maintain stable temperature conditions while compensating for heat loads generated by lighting, equipment operation, or door openings.
4.2 Humidity Control
Relative humidity control is typically achieved through steam injection humidifiers or ultrasonic humidification systems. Dehumidification may be accomplished through refrigeration coils or dedicated moisture removal systems.
Accurate humidity control is essential because moisture levels can significantly influence chemical degradation and physical stability of pharmaceutical products.
4.3 Airflow Distribution
Uniform airflow distribution is necessary to maintain consistent temperature and relative humidity conditions throughout the stability chamber. Circulation fans continuously move conditioned air through the chamber and across storage shelves, promoting uniform exposure of stored samples to the defined environmental conditions.
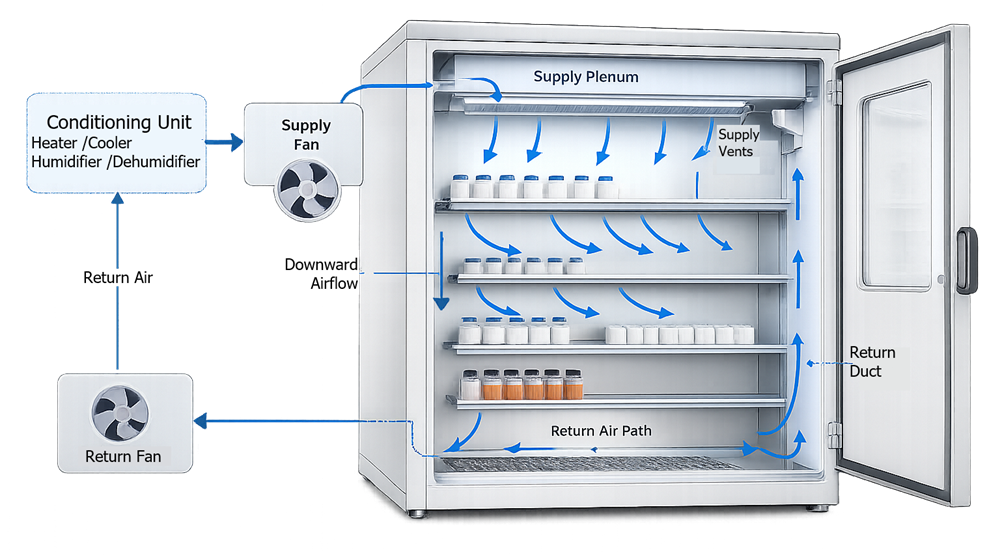
The diagram below illustrates a typical airflow distribution concept used in stability chambers. Conditioned air is typically delivered from a supply plenum located at the top or rear of the chamber and distributed through supply vents into the storage space. The air then flows downward across product shelves and stored samples, providing consistent environmental exposure across all shelf levels.
After passing through the chamber volume, the air returns through return ducts or channels where it is recirculated back to the environmental conditioning system. During this recirculation process, the air may be reheated, cooled, humidified, or dehumidified as required to maintain the defined environmental setpoints.
This continuous recirculation pattern helps maintain stable temperature and humidity conditions throughout the chamber and minimizes localized environmental variations that could affect stability study results.
5. Operational Considerations
Stability chambers are used for long-term studies, and their operation must ensure continuous environmental control throughout the duration of the stability program. Because stability studies may extend for months or years, chamber operation should minimize disturbances that could affect temperature or humidity stability. Typical operational practices include:
- controlled access to minimize door openings
- proper arrangement of samples to allow adequate airflow circulation
- avoidance of blocking air supply or return vents
- routine verification of chamber status and environmental conditions
Opening the chamber door introduces ambient air into the controlled environment and may temporarily disturb both temperature and humidity conditions. The chamber control system must therefore be capable of restoring the defined environmental setpoints within an acceptable recovery period.
5.1 Sample Arrangement and Chamber Loading
The arrangement of stability samples within the chamber plays an important role in maintaining uniform environmental conditions across all storage locations. Samples are typically distributed across shelves with sufficient spacing to allow conditioned air to circulate freely around the stored materials. Air supply vents, return air paths, and circulation fans should remain unobstructed because restricted airflow can create localized temperature or humidity variations within the chamber.
Chamber loading must also be controlled. Excessive loading may interfere with air circulation and reduce the effectiveness of the environmental control system. For this reason, stability chambers are normally operated within loading limits defined by the equipment manufacturer or established during chamber qualification.
During qualification activities, representative loading configurations may be evaluated to demonstrate that temperature and humidity remain uniform throughout the chamber under normal operating conditions.
6. Qualification and Validation Considerations
Because stability studies may extend for months or years, continuous monitoring of environmental conditions is essential. Stability chambers therefore incorporate multiple monitoring sensors and data recording systems to ensure that environmental conditions remain within defined limits throughout the stability study duration. Monitoring systems typically include:
- temperature sensors used for environmental monitoring
- relative humidity sensors used for humidity monitoring
- independent data loggers or chart recorders used for continuous data recording
- centralized monitoring systems connected to facility or laboratory monitoring infrastructure
In many systems, monitoring sensors are independent from the sensors used by the chamber control system. This separation provides an additional layer of verification that environmental conditions remain within specified limits.
Monitoring data are continuously recorded and retained as part of the stability study documentation. These records provide documented evidence that required temperature and humidity conditions were maintained throughout the storage period and support regulatory compliance and data integrity expectations in pharmaceutical stability programs.
6.1 Environmental Monitoring and Data Recording
Because stability studies may extend for months or years, continuous monitoring of environmental conditions is essential. Stability chambers typically incorporate multiple monitoring sensors and data recording systems. Monitoring systems may include:
- temperature sensors used for environmental monitoring
- relative humidity sensors used for humidity monitoring
- independent data loggers or chart recorders
- centralized monitoring systems connected to building management or laboratory monitoring infrastructure
These systems provide continuous documentation of environmental conditions throughout the stability study duration.
6.2 Alarm and Safety Systems
Stability chambers incorporate alarm systems designed to detect deviations from defined environmental conditions. These alarms help ensure that temperature and humidity remain within the limits required for stability studies and allow personnel to respond promptly to abnormal operating conditions. Typical alarms may include:
- high temperature alarms
- low temperature alarms
- high relative humidity alarms
- low relative humidity alarms
- door-ajar alarms indicating prolonged chamber access
- power failure alarms indicating loss of electrical supply
Alarm systems may provide local audible and visual alerts at the chamber as well as remote notifications transmitted to centralized facility monitoring systems or laboratory environmental monitoring platforms.
During equipment qualification, alarm functionality is verified to confirm that alarm setpoints are correctly configured and that notifications are generated when environmental conditions exceed defined limits. This verification demonstrates that deviations from required stability conditions can be detected and addressed in a timely manner.
7. Applications in Pharmaceutical Operations
Stability chambers support regulatory stability programs used to establish product shelf life and storage conditions for pharmaceutical products. These chambers are used to store drug products, drug substances, and packaging configurations under defined environmental conditions for extended periods while periodic analytical testing evaluates product stability. Typical stability programs include:
- long-term stability studies used to establish product shelf life
- accelerated stability studies used to evaluate degradation behavior
- intermediate stability studies used when long-term conditions show instability
- photostability studies evaluating the effect of light exposure
Reliable control of temperature and humidity conditions is essential for generating valid stability data that supports regulatory submissions and product lifecycle management.
