Sterilization Methods Overview
Sterilization is a validated process designed to eliminate all viable microorganisms from a product, component, or system to a defined probability of survival. In pharmaceutical manufacturing, sterility is expressed quantitatively through the Sterility Assurance Level, most commonly 10⁻⁶, representing a maximum probability of one non-sterile unit in one million processed units.
Sterilization processes must be scientifically justified, validated, and maintained in a state of control in accordance with applicable regulatory requirements, including 21 CFR Part 211, EU GMP Annex 1, and applicable ISO standards governing specific sterilization technologies. Method selection must consider product characteristics, material compatibility, process feasibility, and risk to patient safety.
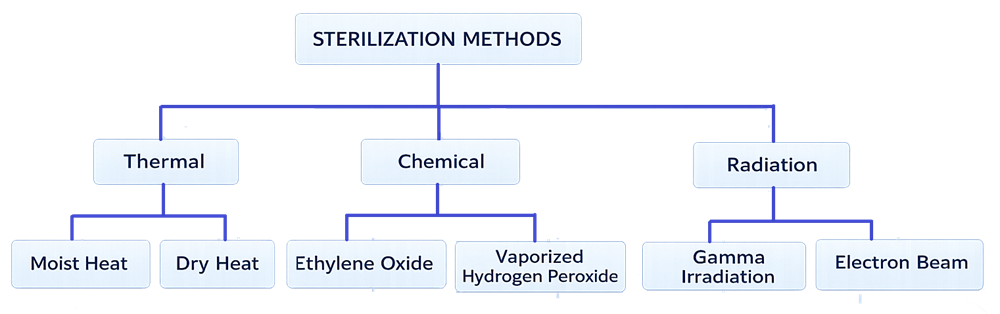
Sterilization technologies used in regulated environments fall into defined categories which can be grouped by primary mechanism of microbial inactivation.
Definitions
Sterilization
A validated process used to eliminate all viable microorganisms from a product, component, or system to a defined probability of survival.
Sterility Assurance Level (SAL)
The probability of a single viable microorganism occurring on an item after sterilization. In pharmaceutical manufacturing, an SAL of 10⁻⁶ is typically required for terminal sterilization processes.
Terminal Sterilization
Sterilization of a product in its final sealed container, providing the highest level of sterility assurance.
Aseptic Processing
Manufacture of sterile product through sterile filtration and filling in controlled environments when terminal sterilization is not feasible.
Moist Heat Sterilization
Moist heat sterilization utilizes saturated steam under pressure to achieve microbial inactivation through protein denaturation. Lethality is typically expressed as F₀, representing equivalent exposure at 121 °C.
This method is considered the preferred sterilization approach when product and material compatibility allow. It is commonly applied to terminally sterilized aqueous products, components, and equipment.
Validation requires:
- Temperature distribution and penetration studies
- Biological indicator qualification
- Lethality calculations
- Load configuration justification
Moist heat remains the regulatory benchmark for robust sterilization performance.
Dry Heat Sterilization and Depyrogenation
Dry heat sterilization employs elevated temperatures without moisture. Microbial inactivation occurs through oxidative damage and dehydration mechanisms.
Applications include:
- Glassware sterilization
- Metal components
- Depyrogenation of containers and closures
Depyrogenation processes must demonstrate validated endotoxin reduction, typically ≥ 3 log reduction for pyrogen control.
Validation includes:
- Temperature mapping
- Endotoxin challenge studies
- Biological indicator confirmation
- Airflow and heat uniformity assessment
Ethylene Oxide Sterilization
Ethylene oxide sterilization is a low-temperature chemical method suitable for heat- and moisture-sensitive materials. The process depends on controlled exposure to EO gas under defined humidity, temperature, and pressure conditions.
EO sterilization is governed by ISO 11135 and requires extensive control of residual gas levels to ensure product safety.
Validation includes:
- Biological indicator resistance verification
- Gas concentration and humidity monitoring
- Aeration studies
- Residual analysis per regulatory limits
Vaporized Hydrogen Peroxide
Vaporized hydrogen peroxide sterilization is widely used for isolators, RABS, and enclosed systems requiring surface decontamination at low temperatures.
Cycle development must demonstrate:
- Uniform vapor distribution
- Target concentration achievement
- Adequate dwell time
- Effective aeration
Biological indicator placement studies are critical to confirm worst-case location lethality.
Radiation Sterilization
Radiation sterilization, including gamma irradiation and electron beam, achieves microbial inactivation through ionizing energy that damages cellular DNA.
It is commonly applied to disposable medical devices and pre-packaged sterile components. These processes are typically performed by qualified external providers.
Validation requires:
- Dose mapping
- Bioburden determination
- Establishment of minimum and maximum absorbed dose
- Material compatibility assessment
Radiation sterilization is governed by ISO 11137.
Regulatory Framework
Sterilization processes used in pharmaceutical manufacturing are governed by regulatory expectations that require scientific validation, documented evidence, and lifecycle control. Key regulatory and guidance references include:
- 21 CFR Part 211 — Current Good Manufacturing Practice
- EU GMP Annex 1 — Manufacture of Sterile Medicinal Products
- ISO 11137 — Radiation Sterilization
- ISO 11135 — Ethylene Oxide Sterilization
- ISO 17665 — Moist Heat Sterilization
- USP guidance related to sterility and endotoxin control
Regulatory authorities consistently expect sterilization processes to be validated, monitored, periodically reviewed, and maintained in a state of control.
Method Selection and Regulatory Preference
Regulatory authorities consistently favor terminal sterilization methods over aseptic processing whenever product characteristics permit. Terminal sterilization provides a higher level of sterility assurance due to treatment of the final sealed container.
Terminal sterilization and aseptic processing differ fundamentally in sterility assurance approach, regulatory preference, and process risk profile.

Selection of a sterilization method must be justified through risk assessment and supported by validation data demonstrating consistent achievement of the required Sterility Assurance Level. Where terminal sterilization is not feasible, aseptic processing must be scientifically justified and supported by enhanced environmental and process controls.
Lifecycle Control
Sterilization validation does not conclude at initial qualification. Ongoing lifecycle management includes:
- Routine monitoring
- Periodic requalification
- Change control evaluation
- Trending of critical parameters
- Review of biological indicator performance
Maintaining a validated state requires structured oversight and documented evidence aligned with regulatory expectations.
