Tablet Compression Equipment
Tablet compression is the final critical unit operation in the manufacture of solid oral dosage forms. During this step, dried and blended granules are compressed into tablets of defined weight, hardness, thickness, and content uniformity. The compression process transforms free-flowing powder or granules into a mechanically stable dosage form suitable for packaging, distribution, and patient use.
Tablet compression equipment must apply controlled mechanical forces to compact the formulation while maintaining uniform tablet properties across the batch. Equipment design, tooling configuration, and process parameters directly influence tablet density, mechanical strength, dissolution performance, and appearance. Because compression occurs at high speeds and involves thousands to millions of tablets per batch, tablet presses must operate with high precision and reliability.
Tablet compression equipment must also support controlled feeding of granules, accurate die filling, consistent compression forces, and effective tablet ejection. Proper qualification and validation ensure that the press consistently produces tablets meeting predefined quality specifications.
1. Purpose of Tablet Compression
The purpose of tablet compression is to convert granules or powders into tablets with consistent physical and chemical characteristics. The compression step must ensure:
- Uniform tablet weight
- Adequate mechanical strength
- Consistent thickness and diameter
- Uniform distribution of active pharmaceutical ingredient
- Acceptable appearance and surface finish
Tablet compression also determines several critical product attributes including dissolution behavior, friability, and mechanical stability during handling. The compression process must therefore be carefully controlled to ensure reproducible tablet formation throughout the batch.
2. Tablet Compression Process Overview
Tablet compression involves several sequential mechanical operations performed by the tablet press. These steps include:
- Granule feeding
- Die filling
- Pre-compression
- Main compression
- Tablet ejection
- Tablet discharge
The illustration below shows the sequence of operations during tablet compression, including granule feeding, die filling, pre-compression, main compression, tablet ejection, and tablet discharge.
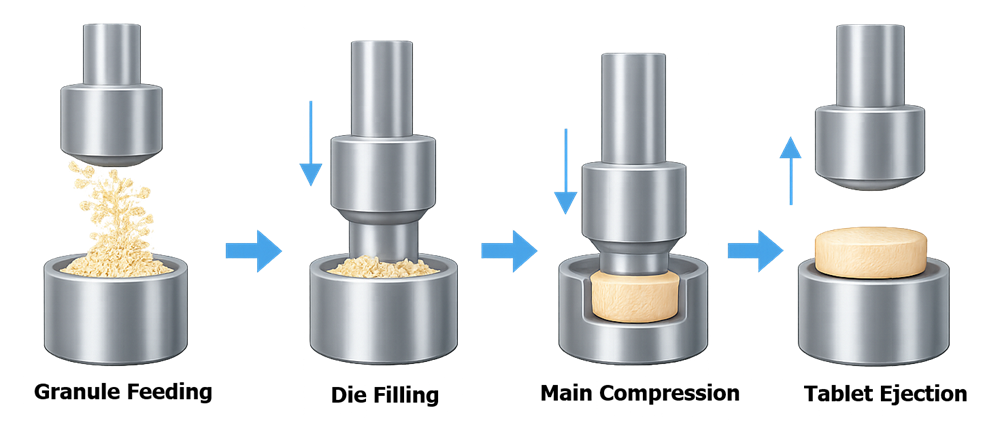
Granules are first transferred from a hopper into the feed frame where rotating paddles distribute the material across the die table. As the turret rotates, dies pass beneath the feed frame and are filled with granules.
Upper and lower punches then apply compression forces that compact the granules into a solid tablet. After compression, the upper punch retracts while the lower punch rises to eject the finished tablet from the die cavity.
The tablet is then removed by a take-off blade and transported for downstream inspection or coating.
3. Types of Tablet Presses
Tablet presses used in pharmaceutical manufacturing fall into two main categories.
3.1 Single-Station Tablet Presses
Single-station presses compress one tablet at a time using a single die and punch set. These presses are typically used for:
- Research and development
- Formulation development
- Small-scale production
Although simple in design, single-station presses allow precise control of compression force and dwell time, making them useful during process development.
The illustration below shows a typical single-station tablet press, often referred to as an eccentric tablet press. In this type of equipment a single set of punches and a die compress one tablet at a time. The upper punch applies compression force while the lower punch supports the granules inside the die cavity and later rises to eject the finished tablet. Single-station presses are commonly used for formulation development, small-scale production, and compression studies because they allow precise adjustment of compression parameters.

3.2 Rotary Tablet Presses
Rotary tablet presses are the most commonly used systems in commercial pharmaceutical manufacturing.
A rotary press contains multiple punch and die sets arranged around a rotating turret. As the turret rotates, each station performs a step in the compression cycle. Rotary presses can produce tens of thousands to hundreds of thousands of tablets per hour depending on machine design and tooling configuration. Advantages of rotary presses include:
- High production capacity
- Consistent compression forces
- Continuous operation
- Integration with automated process monitoring
The illustration below shows a typical rotary tablet press used in pharmaceutical manufacturing. The press contains multiple punch and die stations mounted on a rotating turret that performs sequential operations including die filling, compression, and tablet ejection as the turret rotates.

4. Major Components of a Tablet Press
Tablet presses consist of several key mechanical assemblies that control granule flow, compression forces, and tablet formation.
4.1 Hand Feeding System
Granules are stored in the hopper and delivered to the feed frame where rotating paddles distribute material into the die cavities. The feed frame ensures uniform die filling and prevents segregation of formulation components.
4.2 Turret and Die Table
The turret is the rotating platform that holds multiple dies and punch sets. As the turret rotates, dies pass through the different stages of the compression cycle including filling, compression, and ejection.
4.3 Punches and Dies (Tooling)
Punches and dies form the compression tooling that shapes the tablet. Each tooling set consists of:
- Upper punch
- Lower punch
- Die cavity
The die determines the tablet diameter and thickness while the punches determine the tablet shape and imprint. Tooling geometry directly affects tablet hardness, surface finish, and compression characteristics.
The illustration below shows a typical punch and die tooling set used in tablet compression. The tooling consists of an upper punch, lower punch, and die cavity that define the shape, diameter, and thickness of the tablet.

4.4 Compression Rollers
Compression rollers apply the mechanical force required to compact the granules. Most presses use two compression stages:
- Pre-compression roller
Main compression roller
Pre-compression removes trapped air and improves granule consolidation. Main compression applies the final force required to form the tablet.
The illustration below shows the two compression stages in a rotary tablet press: pre-compression and main compression. As the turret rotates, punch heads pass beneath compression rollers that apply controlled downward force to the upper punch. This force compresses the granules contained in the die cavity between the upper and lower punches.
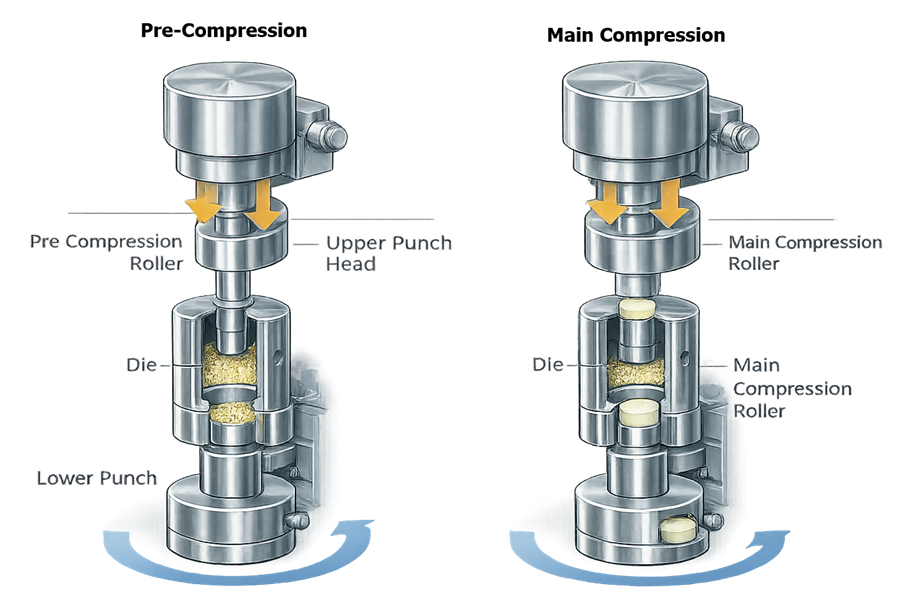
The left side of the illustration represents the pre-compression stage, where a smaller compression force is applied to the granules. Pre-compression removes trapped air from the powder bed and begins initial particle consolidation. This step improves granule packing and helps prevent tablet defects such as capping or lamination during the main compression stage.
The right side of the illustration represents the main compression stage, where a much larger force is applied by the main compression roller. At this stage the granules are compacted into the final tablet structure with the required hardness, thickness, and mechanical strength. After the compression force is released, the upper punch retracts and the lower punch rises to eject the finished tablet from the die cavity.
This sequence ensures controlled densification of the powder bed and consistent tablet formation across all compression stations as the turret rotates.
4.5 Ejection System
After compression, the lower punch rises to push the finished tablet out of the die cavity. A take-off blade guides the tablet away from the die table and transfers it to downstream equipment such as tablet dedusters or metal detectors.
5. Critical Design Features
Tablet compression equipment must incorporate design features that support precise control of the compression process. Important design characteristics include:
- Accurate die filling and feed frame design
- Consistent compression force application
- Minimal vibration during turret rotation
- Precise punch alignment
- Wear-resistant tooling materials
- Dust containment and product collection systems
Equipment must also provide operator safety features and allow easy access for cleaning and maintenance.
6. Process Monitoring and Control
Modern tablet presses incorporate advanced instrumentation and control systems to monitor critical process parameters. Key parameters monitored during compression include:
- Tablet weight
- Compression force
- Pre-compression force
- Tablet thickness
- Turret speed
- Granule feed rate
Automated control systems can adjust press settings in real time to maintain tablet quality within specification limits. Real-time monitoring allows early detection of compression issues such as weight variability, capping, or lamination.
The illustration below shows common mechanical defects that may occur during tablet compression, including capping, lamination, chipping, and sticking. These defects are often associated with improper compression force, formulation issues, or tooling problems.

7. Qualification of Tablet Compression Equipment
Tablet compression equipment must undergo formal qualification to verify that the system is properly installed, operates within defined limits, and consistently produces tablets meeting quality specifications. Qualification typically includes:
- Design Qualification
- Installation Qualification
- Operational Qualification
- Performance Qualification
7.1 Design Qualification
Design Qualification verifies that the selected tablet press meets the requirements defined in the User Requirements Specification. Evaluation typically includes:
- Press capacity and production rate
- Tooling compatibility
- Compression force capability
- Feed frame design
- Control system functionality
- Materials of construction and surface finish
Design Qualification also confirms that safety features and dust containment systems meet regulatory and operational requirements.
7.2 Installation Qualification
Installation Qualification verifies that the tablet press has been installed according to manufacturer specifications and approved engineering documentation. IQ activities typically include:
- Verification of equipment identification and model number
- Confirmation of mechanical installation
- Verification of electrical connections
- Calibration of instrumentation
- Verification of utility connections
- Documentation of equipment manuals and drawings
7.3 Operational Qualification
Operational Qualification verifies that the tablet press operates correctly across its defined operating ranges. Testing typically includes:
- Turret speed control
- Feed frame operation
- Compression force adjustment
- Punch and die movement
- Control system alarms and interlocks
- Emergency stop systems
Operational tests demonstrate that the press functions reliably under controlled operating conditions.
7.4 Performance Qualification
Performance Qualification demonstrates that the tablet compression process consistently produces tablets meeting predefined product specifications during routine manufacturing conditions.
PQ is typically executed using production-scale batches processed under validated operating parameters. PQ studies typically evaluate:
- Tablet weight uniformity
- Tablet hardness
- Tablet thickness
- Tablet friability
- Content uniformity of active ingredient
- Tablet appearance and defect rates
Multiple PQ batches are evaluated to confirm reproducibility and process stability.
7.4.1 Sampling Strategy
Sampling during PQ should represent tablets produced throughout the compression run. Samples are typically collected at the beginning, middle, and end of the batch to evaluate potential variability during operation.
The illustration below shows typical tablet sampling locations and intervals used during tablet press Performance Qualification. Samples are collected at different time points throughout the compression run to verify consistent tablet weight and physical properties.
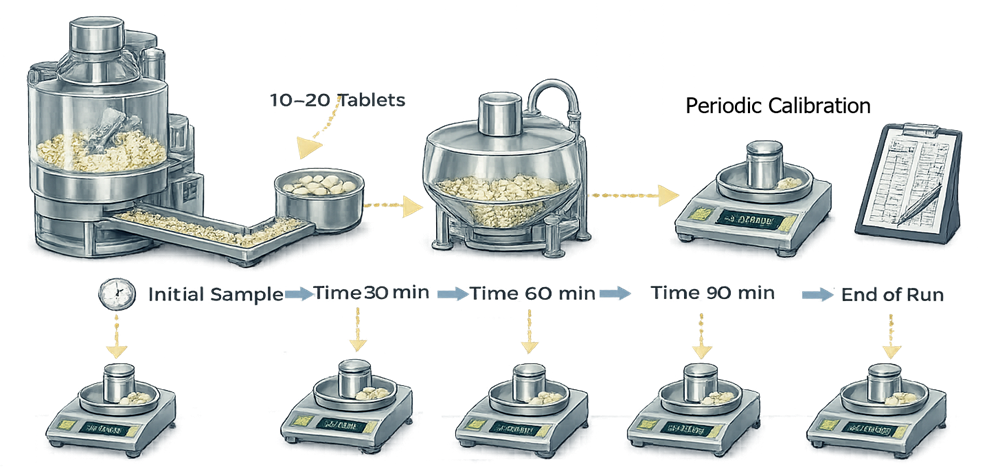
Additional samples may be collected from multiple compression stations to verify uniform tablet formation across the turret.
7.4.2 Statistical Evaluation
Statistical evaluation of PQ data is used to confirm that tablet properties remain consistent across the batch and between batches. Common statistical measures include:
- Mean values
- Standard deviation
- Relative standard deviation
- Trend evaluation across the compression run
These analyses confirm that compression parameters produce tablets within the validated operating range.
7.4.3 Acceptance Criteria
Acceptance criteria must be established before PQ execution. Typical criteria include:
- Tablet weight within specification limits
- Tablet hardness within defined range
- Acceptable friability results
- Content uniformity meeting pharmacopeial requirements
- Absence of visual defects such as capping, lamination, or chipping
Successful completion of Performance Qualification confirms that the tablet press consistently produces tablets that meet product quality specifications.
8. Integration with Downstream Processing
Tablet compression equipment operates as part of the broader solid dosage manufacturing process.
The illustration below shows how tablet compression equipment integrates into the solid dosage manufacturing process. Granules produced during blending are compressed into tablets and subsequently transferred to dedusting, metal detection, coating, and packaging operations.

Granules produced during granulation, drying, and blending are transferred to the tablet press where tablets are formed. After compression, tablets are typically transferred to:
- Tablet dedusters
- Metal detectors
- Tablet coating systems
- Packaging lines
Proper integration between blending, compression, inspection, and packaging systems ensures consistent product quality and efficient manufacturing operations.
Tablet compression therefore represents a critical step in pharmaceutical solid dosage manufacturing and must be carefully controlled to ensure reliable production of high-quality tablets.
