Tank Qualification
1. Introduction
Process vessels used in pharmaceutical manufacturing must be qualified to demonstrate that they are properly installed, operate as intended, and consistently support the manufacturing process. Tank qualification verifies that vessel design, construction, instrumentation, and associated utilities meet defined user requirements and regulatory expectations.
Qualification activities confirm that the vessel can operate safely within specified limits while maintaining hygienic conditions and supporting reliable manufacturing operations. Tank qualification typically follows the standard equipment qualification lifecycle including Installation Qualification, Operational Qualification, and Performance Qualification.
The illustration presents the typical qualification lifecycle used to verify pharmaceutical process vessels before routine manufacturing use.
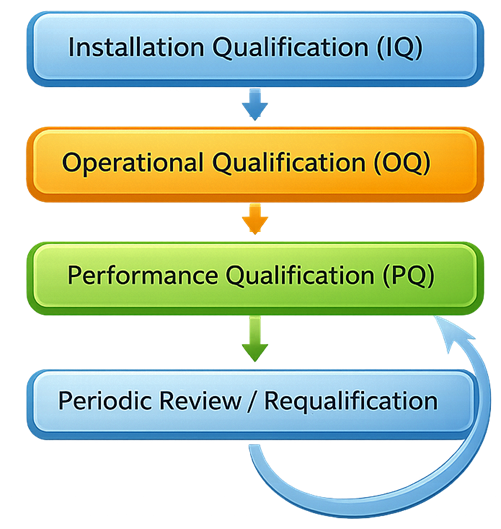
2. Installation Qualification (IQ)
Installation Qualification verifies that the tank has been installed according to approved engineering specifications and manufacturer recommendations.
The illustration below shows typical verification points evaluated during Installation Qualification of a pharmaceutical process vessel. Key elements include materials of construction, weld quality, sanitary process connections, instrumentation installation, and utility interfaces. These verification points confirm that the vessel has been installed according to engineering specifications and design documentation.

Typical IQ activities include verification of:
• equipment identification and asset documentation
• materials of construction and surface finish certifications
• vessel fabrication documentation and weld records
• installation location and structural supports
• utility connections including CIP, SIP, heating or cooling systems
• instrumentation installation and calibration status
• piping connections and sanitary fittings
• safety devices such as pressure relief valves or vacuum breakers
Engineering drawings, piping diagrams, and equipment manuals are typically reviewed during this phase to confirm that the installed system matches the approved design.
3. Operational Qualification (OQ)
Operational Qualification demonstrates that the vessel and its associated systems operate correctly across defined operating ranges.
The illustration shows typical systems evaluated during Operational Qualification of a pharmaceutical process vessel. The diagram highlights the main functional components tested during OQ including agitator operation, temperature monitoring, level measurement, and valve functionality.
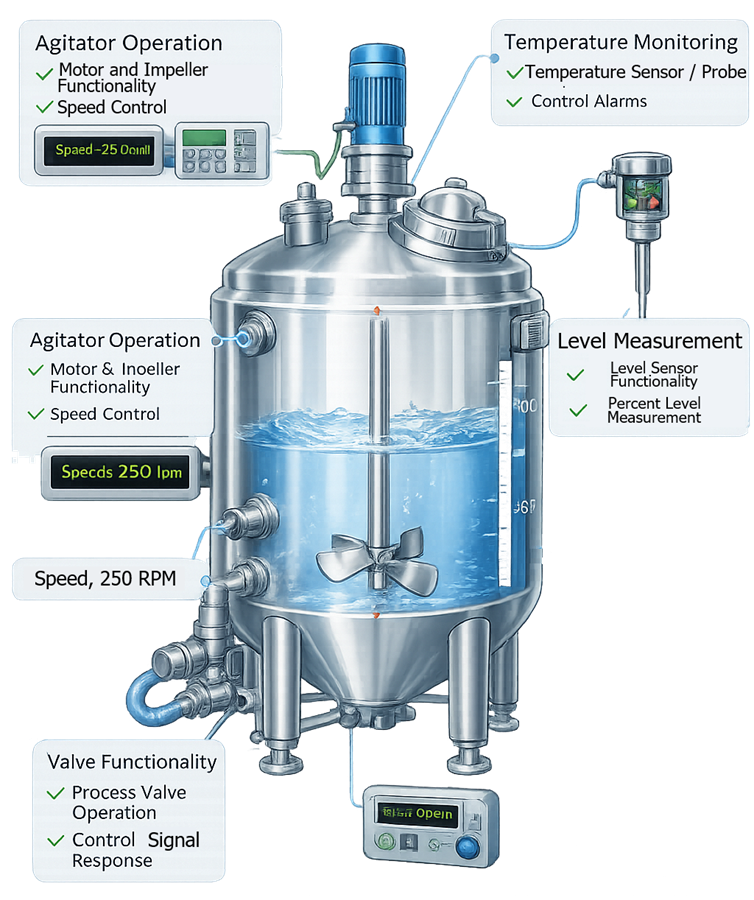
Typical OQ testing may include:
• verification of agitator operation and speed control
• functional testing of temperature control systems
• verification of level measurement devices
• testing of pressure and vacuum protection devices
• functionality of process valves and transfer connections
• testing of CIP spray device coverage and operation
Operational testing ensures that instrumentation, mechanical components, and control systems function correctly under expected operating conditions.
4. Cleanability and CIP Verification
For tanks that are cleaned using automated cleaning systems, qualification activities typically include verification of cleaning performance. Testing may include:
• verification of spray device operation
• coverage testing to confirm cleaning solution reaches all internal surfaces
• verification of drainage following cleaning cycles
• confirmation that CIP parameters operate within defined limits
These activities help demonstrate that the vessel can be effectively cleaned between manufacturing batches. The illustration shows how cleaning solution is distributed inside the vessel during clean-in-place operations.
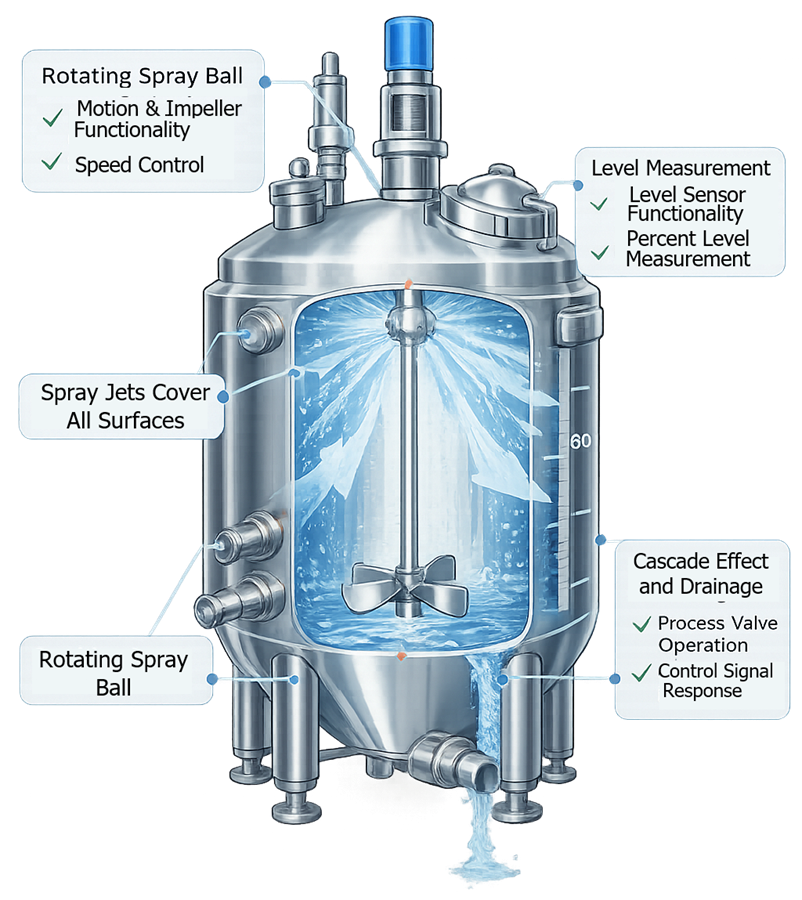
5. Sterilization Capability (When Applicable)
Certain pharmaceutical tanks may be sterilized using steam-in-place systems. When sterilization capability is required, qualification may include verification of sterilization functionality. Typical activities may include:
• verification of steam distribution within the vessel
• monitoring of temperature during sterilization cycles
• verification of condensate removal
• confirmation of sterilization hold times
These tests demonstrate that the vessel can achieve and maintain sterilization conditions when required. The illustration below shows steam distribution inside a process vessel during sterilization operations.

6. Performance Qualification (PQ)
Performance Qualification demonstrates that the tank performs effectively during actual manufacturing operations.
PQ activities may include:
• verification of mixing performance
• confirmation of temperature control stability
• verification of product transfer operations
• evaluation of drainage and product recovery
• monitoring of process parameters during production runs
These activities confirm that the vessel supports consistent process performance under normal manufacturing conditions. The illustration below shows process parameters typically monitored during performance qualification of a process vessel.

7. Documentation and Qualification Records
Tank qualification activities must be documented in accordance with established validation procedures. Qualification documentation typically includes:
• approved qualification protocols
• test records and measurement data
• deviation and corrective action documentation
• final qualification reports
These records demonstrate that the vessel has been evaluated and approved for use in pharmaceutical manufacturing operations.
