Tank Types and Materials
1. Role of Tanks and Process Vessels in Pharmaceutical Manufacturing
Tanks and process vessels are fundamental components of pharmaceutical and biotechnology manufacturing systems. They are used for storage, mixing, reaction, holding, and transfer of process materials including raw ingredients, intermediates, buffers, media, and finished solutions.
The design of these vessels must support hygienic processing, consistent process performance, and effective cleaning. In regulated manufacturing environments, vessel construction, materials, surface finishes, and fabrication practices must be suitable for pharmaceutical use and compatible with applicable regulatory expectations.
Selection of vessel type and material of construction depends on the specific process application, chemical compatibility, operating conditions, and cleaning or sterilization requirements.
2. Tank Categories Based on Process Function
The illustration presents the primary categories of process vessels used in pharmaceutical manufacturing based on their operational function. These include storage tanks, mixing vessels, reactor vessels, buffer or media preparation vessels, and hold vessels. Each vessel type supports a specific stage of material handling or processing within pharmaceutical production systems.

2.1 Storage Tanks
Storage tanks are used to hold raw materials, purified water, buffer solutions, or finished liquid products prior to use or distribution. These tanks typically operate without agitation and are designed to maintain product integrity during storage. Key design considerations include:
- Protection from contamination through sterile vent filtration
- Proper drainability
- Temperature control where required
- Level monitoring and alarm systems
In pharmaceutical water systems, storage tanks often serve as distribution system reservoirs and must support continuous circulation and sanitary operation.
This photograph shows a typical stainless steel storage vessel used in pharmaceutical manufacturing environments. Such tanks are designed to safely hold process liquids while maintaining hygienic conditions and protecting the material from environmental contamination. They are commonly equipped with sanitary process connections, vent filtration, instrumentation ports, and bottom outlet valves that allow controlled transfer of stored liquids to downstream processing equipment.

2.2 Mixing and Formulation Vessels
Mixing vessels are designed to combine multiple components into a homogeneous solution or suspension. These vessels are typically equipped with agitation systems such as impellers, turbines, or magnetic mixers. Important design features include:
- Proper impeller selection to achieve adequate mixing
- Baffle installation to prevent vortex formation
- Instrumentation for temperature, pH, conductivity, or other process parameters
- Capability for controlled ingredient addition
Mixing performance may be critical for ensuring product uniformity, particularly in formulation processes where active pharmaceutical ingredients must be evenly distributed.
This photograph shows a stainless steel mixing vessel used for preparation and formulation of pharmaceutical solutions and suspensions. Multiple sanitary ports allow addition of materials, installation of process instrumentation, and connection to downstream equipment while maintaining hygienic processing conditions.

Mixing vessels contain multiple mechanical and instrumentation components that support controlled blending of pharmaceutical materials. The vessel is equipped with a top-mounted agitator that drives internal mixing elements to promote uniform blending of ingredients. The diagram illustrates typical vessel features including agitation systems, baffles, sampling ports, and bottom drain connections.
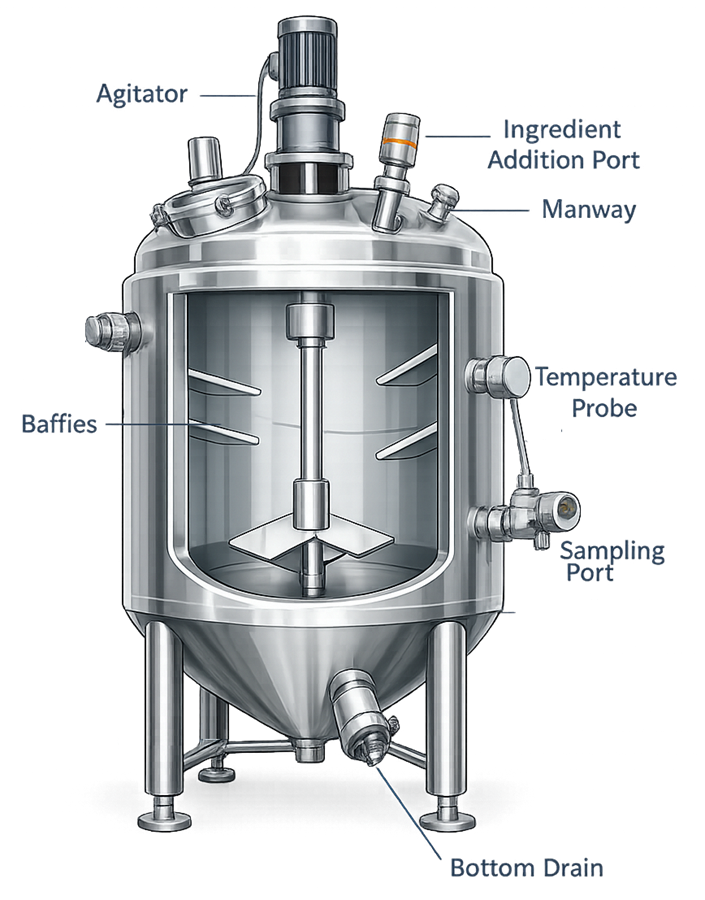
2.3 Reactor Vessels
RReactor vessels are used for chemical or biochemical reactions requiring controlled process conditions. These vessels often incorporate thermal control systems, advanced agitation mechanisms, and multiple instrumentation ports that allow precise monitoring and regulation of reaction parameters.
Typical reactor vessel capabilities include:
- Temperature control through external jackets or internal heating and cooling coils
- Operation under controlled pressure or vacuum conditions
- Controlled reagent or ingredient addition through dedicated ports
- Sampling ports for monitoring reaction progress and product characteristics
Reactor vessels are widely used in active pharmaceutical ingredient manufacturing as well as in certain biotechnology and biochemical processing applications where controlled reaction environments are required.
The illustration below presents the configuration of a typical jacketed reactor vessel. The diagram shows the principal components that support controlled processing operations. A top-mounted agitator drives internal mixing blades to ensure uniform distribution of reactants and heat within the vessel contents.
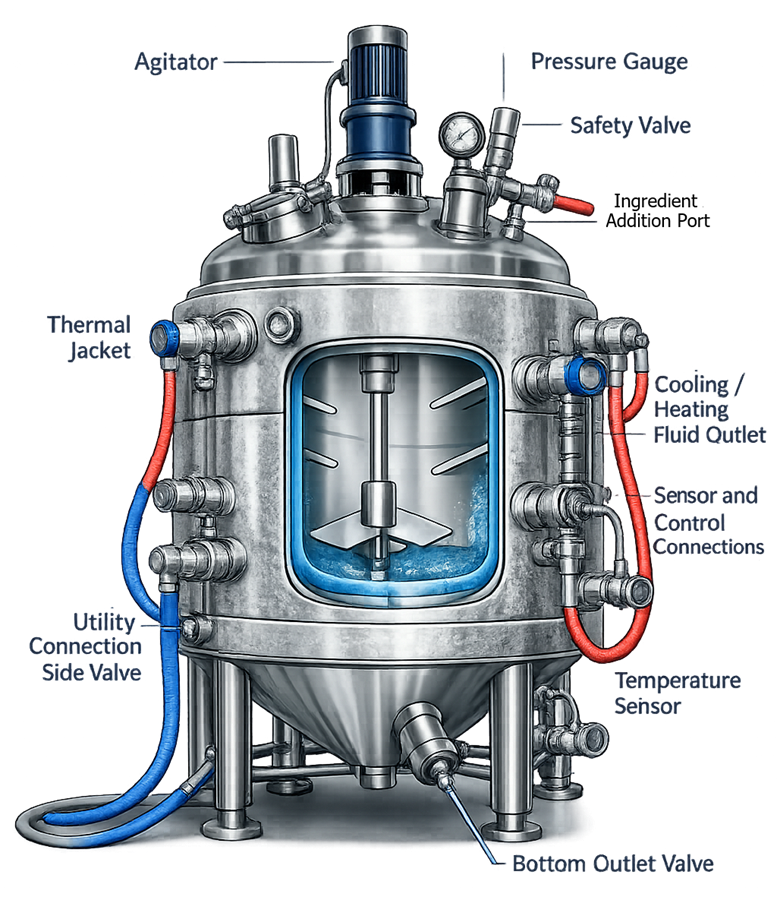
Surrounding the vessel wall is a thermal jacket that allows circulation of heating or cooling media. Dedicated inlet and outlet connections permit controlled flow of thermal fluid through the jacket, enabling precise temperature regulation of the process contents.
Additional vessel head connections support installation of process instrumentation such as pressure gauges and temperature sensors, as well as safety devices including relief valves. Ingredient addition ports allow controlled introduction of reagents during the reaction process.
The vessel also includes multiple utility connections and a bottom outlet valve that allows controlled discharge of the processed material once the reaction step is complete. Together, these design features enable reactor vessels to maintain stable process conditions while supporting safe and controlled pharmaceutical manufacturing operations.
2.4 Buffer and Media Preparation Vessels
In biotechnology and cell culture manufacturing, vessels are frequently used to prepare buffers and culture media. These solutions are used throughout upstream and downstream processing steps. These vessels often include:
- Agitation systems for dissolution of powders
- Load cells or level measurement for volume control
- Filtration interfaces for sterile transfer
- Integration with automated process control systems
Single-use vessels are increasingly used for buffer and media preparation in modern bioprocess facilities.
2.5 Hold Vessels
Hold vessels are used to temporarily store product or intermediates between processing steps. These vessels may maintain sterile conditions, temperature control, or gentle agitation depending on process requirements.
Design considerations include:
- Maintaining product quality during hold time
- Compatibility with cleaning or sterilization processes
- Integration with upstream and downstream process equipment
Hold time limits are typically defined during process validation.
2.6 Portable Tanks and Mobile Vessels
Portable tanks are movable vessels used for temporary storage, staging, or transfer of materials between processing steps. These tanks are commonly mounted on wheeled frames or carts and may be manually transported or moved using material handling equipment. Portable vessels are frequently used for:
- Intermediate product transfer between rooms
- Staging of prepared solutions prior to processing
- Feeding mixers, reactors, or filling equipment
- Buffer or excipient preparation in smaller volumes
Typical design features include:
- Stainless steel sanitary construction
- Lockable wheels or mobile frames
- Bottom outlet valves for controlled transfer
- Sanitary ports for venting or sampling
- Removable lids or sealed covers

Because portable vessels are frequently moved between process areas, additional operational considerations apply:
- Cleaning and sanitization between uses
- Prevention of cross-contamination during transport
- Labeling and identification of vessel contents
- Controlled storage when not in use
Portable tanks must be included in equipment qualification and cleaning validation programs where they contact product or process materials. Their mobility also requires clear procedures for handling, cleaning, and use to maintain GMP compliance.
3. Materials of Construction
3.1 Stainless Steel
Stainless steel is the most widely used material for pharmaceutical process vessels. The preferred grade is 316L stainless steel due to its corrosion resistance, mechanical strength, and compatibility with pharmaceutical cleaning and sterilization procedures. Advantages include:
- Excellent resistance to corrosion
- Compatibility with steam sterilization
- Smooth, cleanable surfaces
- Long service life
Surface finish is an important specification for stainless steel vessels. Pharmaceutical applications typically require internal surface roughness values of approximately Ra ≤ 0.5–0.8 µm, which supports effective cleaning and minimizes microbial attachment.
Material traceability and certification are normally required, including mill test reports and verification of alloy composition.
3.2 Glass-Lined Steel
Glass-lined reactors are sometimes used in chemical synthesis processes, particularly when highly corrosive reagents are present. The glass lining provides chemical resistance while the steel structure provides mechanical strength.
These vessels are most common in active pharmaceutical ingredient production rather than sterile drug product manufacturing.
Design considerations include protection of the glass lining from mechanical damage and careful inspection for cracks or defects.
3.3 High-Purity Plastics
Certain applications utilize high-purity polymer materials such as:
- Polypropylene
- PVDF
- HDPE
These materials may be used in smaller vessels, transfer containers, or specialized systems where chemical compatibility or cost considerations are important. Polymer vessels must be evaluated for extractables and leachables where product contact occurs.
3.4 Single-Use Vessel Systems
Single-use vessels are widely used in modern biologics manufacturing and are typically constructed from multilayer polymer films supported by rigid containers or frames. Advantages include:
- Elimination of cleaning requirements
- Reduced risk of cross-contamination
- Rapid system changeover
- Reduced water and chemical consumption
However single-use systems require evaluation of material compatibility, extractables and leachables, and mechanical integrity during use.

The illustration shows a single-use bioprocess vessel system used in biotechnology and biologics manufacturing. In this design the product is contained inside a disposable multilayer polymer bag supported by a reusable stainless steel frame. The disposable bag forms the product-contact surface and integrates sterile tubing and sensor ports, eliminating the need for vessel cleaning between batches.
A motor-driven agitator mounted on the vessel head drives an internal impeller that provides mixing of the process fluid. Mixing ensures uniform distribution of nutrients, dissolved gases, and added components during processing.
Several process sensors are connected through sterile ports on the disposable bag. These typically include a pH probe for monitoring acidity, a dissolved oxygen probe for measuring oxygen concentration, and a level sensor used to monitor the liquid volume inside the vessel.
The system includes a sterile liquid inlet for addition of media, buffers, or other ingredients. An exhaust filter located at the top of the vessel allows gas exchange while maintaining sterile conditions inside the bag.
Flexible transfer lines connect the vessel to upstream or downstream equipment. A sampling line allows removal of small volumes of process fluid for analytical testing, while a bottom outlet line enables controlled discharge of the processed liquid.
A separate control console monitors sensor signals and controls agitator operation, allowing operators to maintain stable process conditions throughout the bioprocess operation.
4. Surface Finish and Sanitary Construction
Surface finish and fabrication practices play a critical role in sanitary vessel design. Poor surface quality or fabrication defects can create areas where product residues or microorganisms accumulate. Key considerations include:
- Smooth internal surfaces with controlled roughness
- Properly polished welds
- Elimination of crevices and rough areas
- Sanitary connections such as tri-clamp fittings
Weld quality is typically verified through inspection and documentation such as weld maps and boroscope inspection where appropriate.
The photograph below shows the interior of a stainless steel pharmaceutical process vessel with a polished hygienic surface finish. The smooth reflective walls and sanitary weld transitions are designed to minimize surface roughness and prevent accumulation of product residues or microorganisms. Visible components include the central agitator shaft with mixing impeller and multiple spray devices used for clean-in-place operations. Such polished internal surfaces support effective cleaning, drainage, and sterilization, which are critical requirements for pharmaceutical manufacturing equipment.

In addition to surface finish and weld quality, pharmaceutical process vessels incorporate several other hygienic design features that support cleanability, safe operation, and reliable process performance. Vessel heads are often domed or dished, which promotes drainage and prevents accumulation of liquid on the upper surfaces during cleaning or sterilization. Process vessels are equipped with sanitary manways or access ports that allow inspection, maintenance, and installation of internal components while maintaining hygienic sealing during operation.
For automated cleaning, vessels typically include clean-in-place spray devices, such as fixed spray balls or rotary spray heads, positioned to ensure complete coverage of internal surfaces during cleaning cycles.

Process and instrumentation connections are commonly made through tri-clamp sanitary fittings, which provide smooth, crevice-free interfaces that are easy to disassemble and clean when required.

Pharmaceutical tanks also incorporate sanitary vent and pressure ports, which allow pressure equalization and connection of sterile vent filters to protect the vessel contents from environmental contamination. Ingredient addition ports or process inlets enable controlled introduction of materials while maintaining hygienic conditions.

To ensure proper drainage, vessels are normally constructed with sloped or conical bottoms that direct liquid toward a sanitary discharge point.

The product is typically removed through a sanitary bottom outlet valve, which is designed to minimize dead space and allow complete drainage of the vessel contents

Finally, vessels are supported by adjustable hygienic legs or frames, which allow leveling of the equipment and ensure proper drainage during operation and cleaning. These features collectively contribute to the hygienic design and reliable operation of pharmaceutical process vessels.
5. Material Compatibility with Process and Cleaning Conditions
MMaterial selection for pharmaceutical process vessels must consider all chemical, thermal, and mechanical conditions encountered during operation. The materials used for product-contact surfaces must remain stable when exposed to process fluids, cleaning agents, and sterilization conditions. Improper material selection can result in corrosion, degradation, contamination of the product, or reduced equipment lifespan.
One of the primary considerations is corrosion resistance. Process chemicals, buffer solutions, acids, bases, and cleaning agents can attack certain metals if the material is not properly selected. For this reason, 316L stainless steel is commonly used for pharmaceutical vessels due to its resistance to corrosion and compatibility with most pharmaceutical processing environments.
Another important factor is chemical compatibility. Vessel materials must not react with or be degraded by the substances they contain. Incompatible materials may release contaminants, absorb process components, or undergo chemical changes that compromise product quality.
Process vessels must also withstand the temperature and pressure conditions encountered during operation. Heating, cooling, sterilization, or pressurized processing may expose the vessel to elevated temperatures and internal pressures. Materials must maintain mechanical integrity and dimensional stability under these conditions.
Equally important is the requirement to protect product purity. Materials used in pharmaceutical equipment must not introduce extractables, leachables, or particulates into the product stream. This is especially critical for product-contact surfaces where contamination could compromise product safety and regulatory compliance.

The illustration highlights key factors influencing material compatibility in pharmaceutical process vessels, including corrosion resistance, chemical compatibility with process fluids, resistance to operating temperature and pressure conditions, and protection of product purity. Proper evaluation of these factors ensures that vessel materials remain suitable for their intended use throughout the equipment lifecycle.
