Design Requirements for Tanks and Process Vessels
1. Introduction
Pharmaceutical process vessels must be designed to support hygienic operation, reliable process performance, and compliance with Good Manufacturing Practice requirements. The design of tanks and process vessels must allow safe containment of process materials while preventing contamination and enabling effective cleaning and sterilization.
Proper engineering design ensures that vessels operate safely under defined process conditions and maintain product quality throughout the manufacturing process. Design considerations include vessel geometry, hygienic construction, instrumentation, pressure ratings, and integration with utilities such as cleaning and sterilization systems.
2. Sanitary Geometry and Cleanability
The geometry of pharmaceutical process vessels must support complete cleaning and drainage. Poorly designed geometries can create areas where product residues accumulate, increasing the risk of contamination or microbial growth. The illustration below shows a typical hygienic tank geometry designed to support effective cleaning and drainage. The diagram highlights key geometric features commonly incorporated into pharmaceutical process vessels.
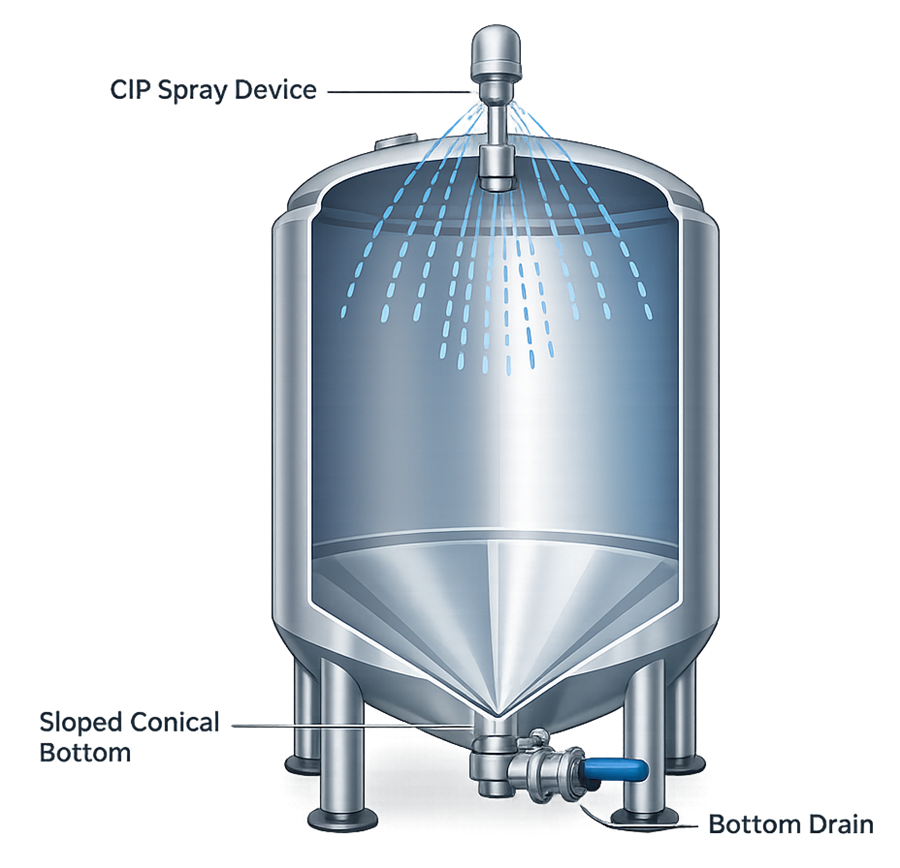
Key hygienic geometry considerations include:
• elimination of dead legs and stagnant zones
• smooth internal surfaces and sanitary connections
• sloped or conical vessel bottoms to promote drainage
• proper positioning of cleaning spray devices
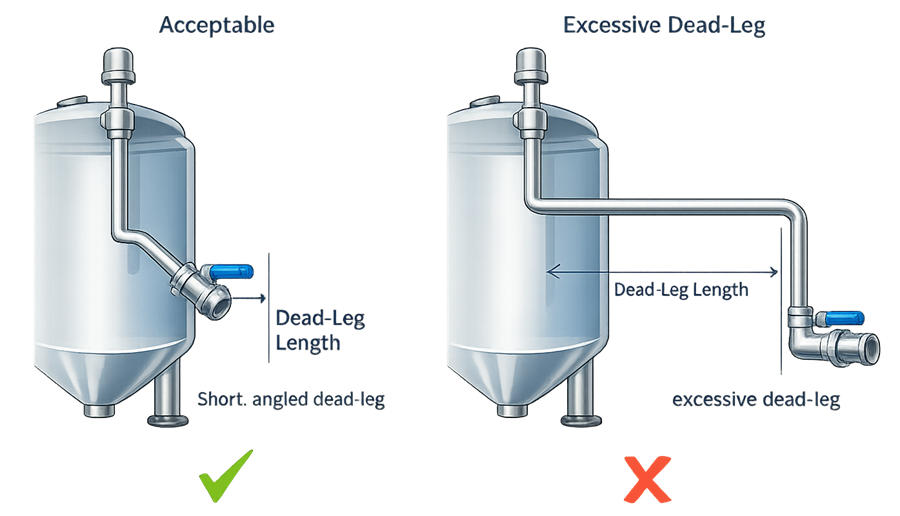
Dead-leg lengths should be minimized to reduce the risk of stagnant fluid retention. Industry practice commonly limits dead-leg length relative to pipe diameter to maintain effective cleaning and fluid circulation. The illustration presents a comparison of piping configurations that create dead legs and configurations designed to minimize stagnant zones in hygienic process equipment.
Drainability is a critical design parameter. Vessel bottoms are typically designed with a conical or sloped geometry that directs liquids toward the bottom outlet valve, allowing complete removal of product and cleaning solutions.
3. Vessel Geometry and Structural Design
The physical geometry of process vessels must support both mechanical stability and process performance. Tanks may be designed in vertical or horizontal configurations depending on space constraints and process requirements. Important structural considerations include:
• vessel diameter and height ratio
• wall thickness and mechanical strength
• support structures and mounting frames
• access ports and manways for inspection
The illustration shows the main structural components of a typical pharmaceutical process vessel and their relative positions within the vessel design.

Process vessels must also be designed to withstand the loads generated by mixing systems, internal pressure, and external environmental conditions.
Manways and access ports allow operators to inspect vessel interiors and perform maintenance activities. These openings must be equipped with sanitary seals to maintain hygienic integrity during operation.
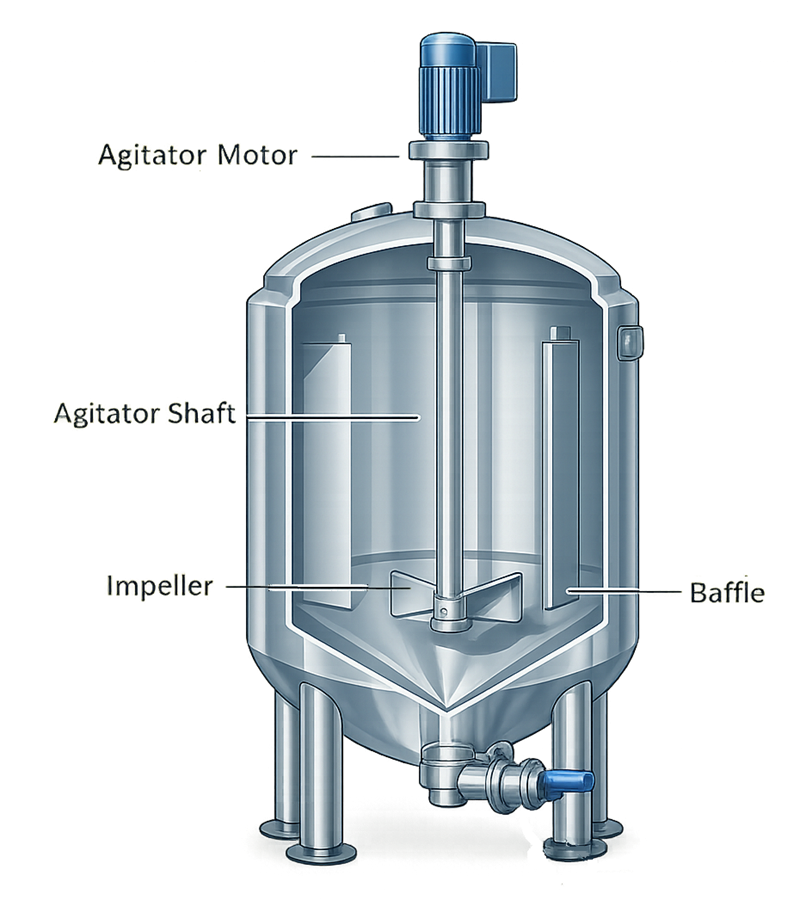
4. Agitation and Mixing Design
Many pharmaceutical process vessels require mixing systems to ensure uniform distribution of process components. Proper mixing design is essential for maintaining consistent product composition and process performance. Agitation systems typically include:
• top-mounted or bottom-mounted agitator drives
• internal impellers or mixing blades
• baffles to prevent vortex formation
The selection of impeller type and mixing speed depends on the viscosity of the process fluid, the required mixing intensity, and the volume of the vessel.
Agitation systems must be designed to provide adequate mixing while minimizing shear forces that could damage sensitive biological materials or degrade product quality. The illustration presents a typical mixing system configuration used in pharmaceutical process vessels.
5. Instrumentation and Process Connections
Pharmaceutical process vessels incorporate instrumentation and process connections that allow monitoring of operating conditions and support controlled manufacturing operations. These connections provide interfaces for measurement of process parameters, material addition, sampling, venting, and product transfer. All instrumentation and connection points must be designed using sanitary process connections that maintain hygienic conditions and support effective cleaning and sterilization.
5.1 Process Monitoring Instrumentation
Instrumentation installed on process vessels allows operators and control systems to monitor critical operating parameters during manufacturing. Typical instrumentation includes:
• Temperature sensors used to monitor product temperature and support temperature control systems
• Pressure gauges or pressure transmitters used to monitor vessel pressure during operation
• Level measurement devices used to determine the volume of liquid within the vessel
• Analytical probes, such as pH sensors or conductivity probes, used to monitor chemical properties of the process fluid
Instrumentation is commonly installed through sanitary instrument ports that allow installation, removal, and calibration without compromising the hygienic integrity of the vessel.
5.2 Thermal Jacket Monitoring
Process vessels equipped with heating or cooling jackets require additional instrumentation to monitor jacket operating conditions. Jacket-side monitoring typically includes:
• Temperature sensors on the jacket supply and return lines
• Pressure gauges or pressure transmitters installed on the jacket circuit
These measurements allow operators to verify proper operation of the heating or cooling system and ensure that the jacket operates within its design limits.
5.3 Process Ports and Operational Connections
In addition to monitoring instrumentation, vessels include several process ports that support manufacturing activities. Common connections include:
• Sampling ports used to withdraw product samples during processing
• Ingredient addition ports used to introduce powders or liquids into the vessel
• Vent ports connected to sterile filters to allow pressure equalization while protecting the vessel contents from environmental contamination
• Transfer connections used to move product to downstream equipment such as filtration systems or holding tanks
These ports must be designed to allow safe and controlled process operations while maintaining hygienic conditions.
5.4 Sanitary Design of Instrument Connections
All instrumentation and process ports must be designed to avoid crevices, stagnant zones, or liquid hold-up areas that could interfere with cleaning or promote microbial contamination. Sanitary design practices include the use of sanitary fittings, proper orientation of instrument ports, and installation methods that support clean-in-place and sterilization procedures.
Proper design and placement of instrumentation and process connections ensure that vessels can be effectively monitored and controlled while maintaining the hygienic requirements of pharmaceutical manufacturing.
6. Pressure and Vacuum Design
Process vessels may operate under atmospheric pressure, positive pressure, or vacuum conditions depending on the manufacturing process. Vessel design must account for these operating conditions to ensure safe operation. Design considerations include:
• vessel pressure ratings
• installation of pressure relief devices
• vacuum breakers or pressure regulators
• structural reinforcement where required
Pressure relief valves protect the vessel from over-pressurization, while vacuum breakers prevent vessel collapse during vacuum conditions or rapid cooling. The illustration presents common safety devices installed on pharmaceutical process vessels to protect against excessive pressure or vacuum conditions.

These protective devices are critical safety features in pharmaceutical process equipment.
7. Utility Integration
Pharmaceutical tanks often integrate with multiple utility systems that support manufacturing operations. These utilities may include heating, cooling, cleaning, and sterilization services. Typical integrated utilities include:
• heating or cooling jackets
• clean-in-place systems
• steam-in-place sterilization systems
• sterile vent filtration
The illustration shows typical utility connections integrated with pharmaceutical tanks to support heating, cooling, cleaning, and sterilization functions.
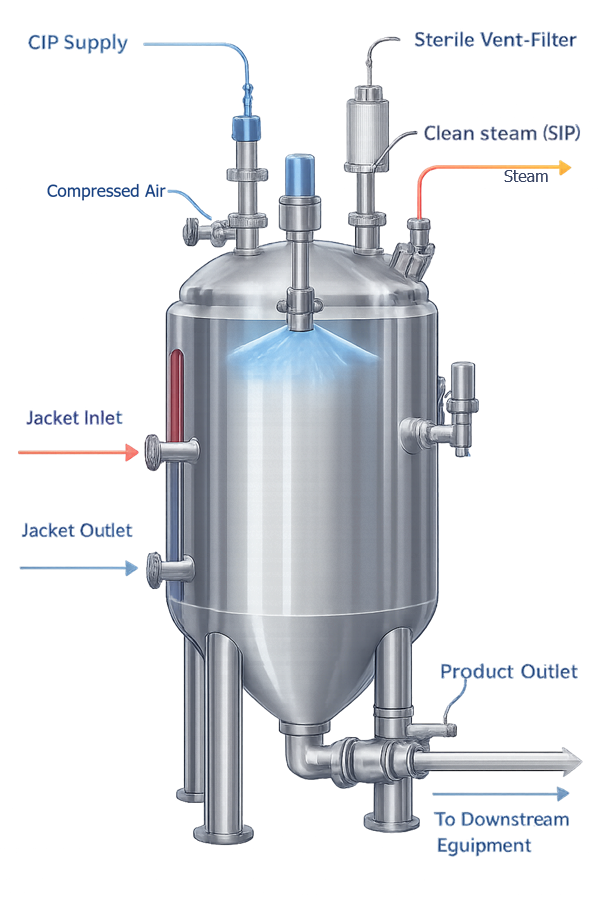
Proper integration of these systems ensures that the vessel can be effectively cleaned, sterilized, and operated within controlled process parameters.
8. Drainage and Bottom Outlet Design
Effective drainage is essential for preventing product hold-up and ensuring complete removal of process fluids. Vessel bottoms are typically designed with conical or sloped geometries that direct liquids toward the lowest discharge point.
The bottom outlet valve must be designed to minimize dead space and allow full evacuation of vessel contents.
Proper outlet design supports:
• efficient product recovery
• effective cleaning and rinsing
• prevention of stagnant fluid accumulation
Drainability is a key requirement for hygienic vessel design and is often verified during equipment qualification and cleaning validation activities.
9. Design Documentation
Engineering documentation is an important component of pharmaceutical equipment design. Documentation ensures that the vessel design meets specified requirements and provides traceability for materials and fabrication processes.
Typical design documentation may include:
• engineering drawings and fabrication diagrams
• material certifications
• weld maps and inspection records
• surface finish documentation
• pressure rating and design calculations
These records support equipment qualification and demonstrate compliance with regulatory expectations for pharmaceutical manufacturing equipment.
