Temperature Mapping and Heat Distribution
Temperature mapping is the technical demonstration that a steam sterilization system achieves uniform and reproducible thermal conditions under defined operating parameters. It identifies the coldest location, verifies exposure conditions, and provides the thermal foundation upon which lethality calculations are based.
Mapping establishes thermal capability. Biological indicators confirm microbiological lethality. Both are required for defensible sterilization validation.
1. Purpose of Temperature Mapping
Temperature mapping studies are performed to:
- Confirm uniform heat distribution within the chamber
- Identify the coldest chamber location
- Verify attainment and maintenance of exposure temperature
- Support F₀ calculation at the worst-case location
- Confirm load development assumptions
- Demonstrate cycle reproducibility
Mapping is a controlled engineering study, not a procedural formality.
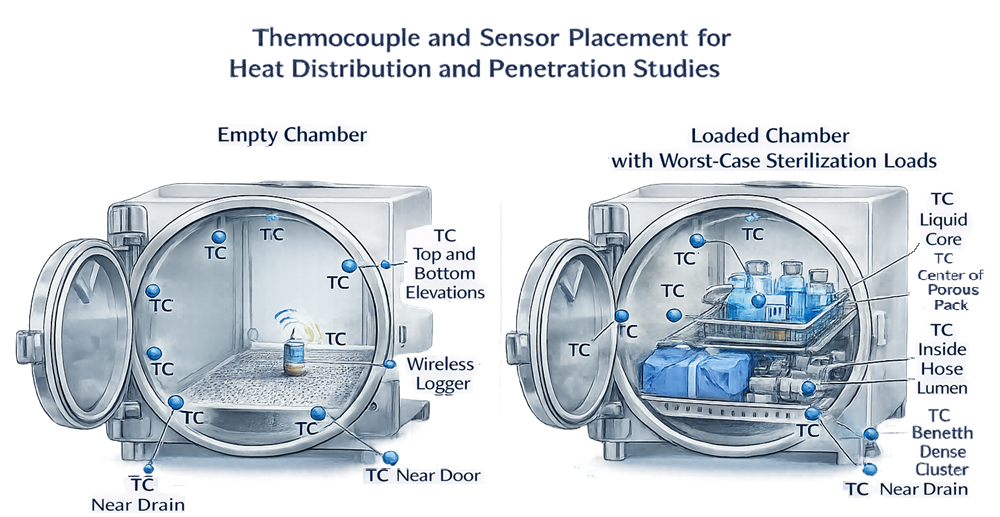
2. Heat Distribution Studies – Empty Chamber
Heat distribution studies are typically executed during Operational Qualification.
Objectives
- Demonstrate chamber temperature uniformity
- Confirm control system stability
- Identify coldest chamber location
- Verify drain temperature behavior
The chamber drain is often, but not always, the coldest point due to condensate accumulation and steam flow dynamics.
Sensor Quantity and Placement
The number of temperature sensors is determined based on:
- Chamber volume
- Chamber geometry
- Regulatory guidance
- Risk assessment
- Historical system performance
Industry standards such as ISO 17665 and established validation practice correlate minimum sensor count with chamber size. Larger chambers require proportionally more measurement points to ensure spatial variability is adequately characterized.
Sensor placement typically includes:
- Chamber corners
- Near the door
- Near the drain
- Upper and lower elevations
- Geometric center
The study protocol must justify sensor count and placement rationale.
3. Heat Penetration Studies – Loaded Chamber
Heat penetration studies confirm thermal performance under worst-case load conditions defined during load development.
Objectives
- Confirm heat transfer into the load
- Identify internal cold spots
- Validate biological indicator placement
- Demonstrate adequate lethality at the slowest-heating location
Loaded mapping verifies that the sterilization cycle is effective not only in the chamber space, but within the product or equipment assembly.
Probe Placement Strategy
Sensor placement within the load is determined through engineering evaluation and documented load development studies.

Example of instrumented equipment assemblies during heat penetration study: Multiple stainless steel vessels fitted with calibrated thermocouples to evaluate internal heat penetration during steam sterilization qualification.

Thermocouple insertion through sanitary fitting during validation study
Close-up of temperature probe installed through sanitary port to monitor internal vessel temperature during steam sterilization qualification.
Typical locations include:
- Center of porous packs
- Core of liquid containers
- Internal lumen of hoses
- Filter housings
- Beneath dense clusters of components
- Most remote location from steam entry
The slowest-heating location defines the worst-case lethality reference point.
4. Temperature Measurement Technologies
Mapping studies in moist heat sterilization may utilize:
- Thermocouples
- Resistance temperature detectors
- Wireless high-temperature data loggers
Thermocouples are traditionally preferred for steam sterilization due to:
- Rapid response time
- Suitability for high-temperature environments
- Integration with validation data acquisition systems
Wireless data loggers may also be used, provided they are:
- Rated for sterilization temperatures
- Calibrated and traceable to national standards
- Validated for accuracy and response characteristics
Regardless of technology, all sensors must:
- Be calibrated prior to use
- Have documented accuracy
- Be traceable to recognized standards
- Be evaluated for placement integrity
Measurement system accuracy directly affects F₀ calculation and lethality determination.
5. Heat Distribution vs Heat Penetration
These two evaluations serve different purposes.
Heat Distribution
Confirms uniform temperature within an empty chamber.
Heat Penetration
Confirms temperature response within a defined load.
A sterilizer may demonstrate excellent chamber distribution yet exhibit delayed penetration within a dense or complex load. Both studies are necessary.
6. Data Evaluation and Acceptance Criteria
Mapping data are evaluated for:
- Time to reach exposure temperature
- Stability during exposure phase
- Maximum temperature variability
- Cold spot identification
- Duration above minimum sterilization temperature
- F₀ achieved at worst-case location
All probes must achieve minimum exposure conditions for the required duration.
Transient deviations must be evaluated in the context of cumulative lethality.
The cycle is validated at the slowest-heating location.
7. Relationship to Qualification
Temperature mapping supports:
- Operational Qualification through empty chamber studies
- Performance Qualification through loaded heat penetration studies
- Biological indicator placement strategy
- Lethality margin justification
Mapping establishes the thermal evidence that the sterilization process is capable. Biological indicators confirm the microbiological outcome.
Without scientifically executed mapping, sterilization validation lacks technical foundation.
