Pharmaceutical Water Quality Categories and Intended Use
Pharmaceutical water is a manufactured GMP material produced on site under controlled conditions. It is governed by pharmacopeial requirements and regulatory expectations in the same manner as other critical raw materials. Water quality selection is not discretionary. It is dictated by intended use, and that decision drives system design, monitoring, qualification, and lifecycle control.
Failure to align water quality with intended use remains a recurring regulatory observation.
Water as a GMP-Controlled Material
Pharmaceutical waters are subject to defined specifications, controlled production, and ongoing verification. As described in USP <1231>, water quality must be appropriate for its application and supported by documented rationale.
Key expectations:
- Intended use is defined and approved
- Appropriate USP water category is selected
- Use does not exceed documented justification
- Control is demonstrated through monitoring and trending
Historical acceptance does not replace documented compliance.
Pharmaceutical Water Quality Categories
USP defines multiple water categories, each with a specific role and boundary of use.
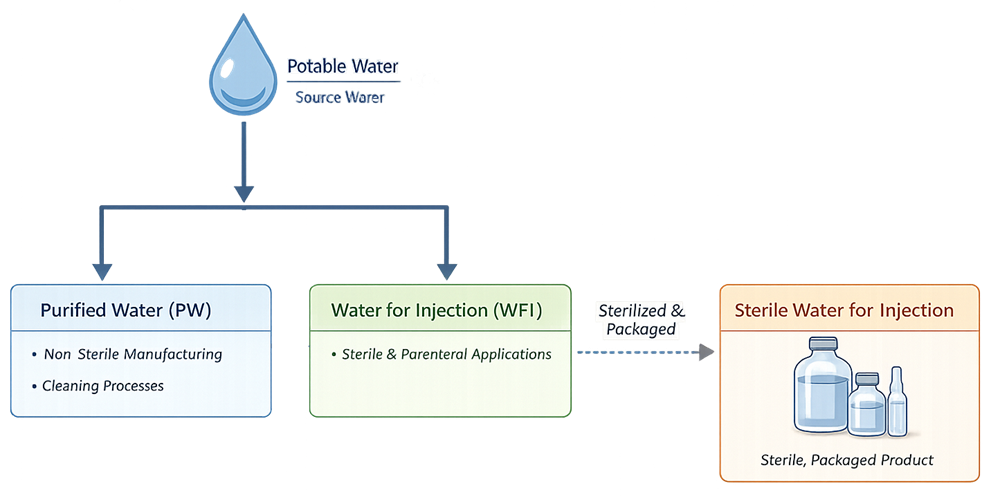
Pharmaceutical water categories are selected strictly based on intended use. Purified Water and Water for Injection are generated and distributed on site as GMP utilities and are controlled through continuous system operation, monitoring, and trending. Sterile Water for Injection is derived from Water for Injection, then sterilized and packaged as a finished pharmaceutical product. Once packaged, it is no longer managed or distributed as a utility and is controlled through batch release.
Potable Water
USP Reference: USP <1231> Water for Pharmaceutical Purposes
Potable water is the required feed source for pharmaceutical water systems.
Characteristics:
- Meets local drinking water regulations
- Used only as system feed water
- Not acceptable for direct product contact or final equipment rinsing
Potable water quality directly impacts pretreatment design, fouling potential, and long-term system performance.
Purified Water (PW)
USP References:
- Purified Water monograph
- USP <1231> Water for Pharmaceutical Purposes
Purified Water is the most widely used pharmaceutical water grade.
Typical intended uses:
- Non-sterile drug product manufacturing
- Equipment cleaning and rinsing
- Component washing
- Non-sterile compounding activities
USP expectations:
- Compliance with chemical purity requirements
- Microbiological control
- Endotoxin control only when justified by use
Purified Water systems are considered direct-impact GMP systems and require formal qualification and routine performance monitoring.
Water for Injection (WFI)
USP References:
- Water for Injection monograph
- USP <1231> Water for Pharmaceutical Purposes
Water for Injection is required when endotoxin control is mandatory and enhanced microbiological control is required by intended use.
Typical intended uses:
- Parenteral drug product manufacturing
- Aseptic processing operations
- Final rinse for equipment used in sterile manufacturing
USP expectations:
- Stringent chemical purity
- Defined microbiological limits
- Endotoxin control
- Continuous circulation and effective sanitization
WFI systems are high-risk utilities and are routinely examined in depth during regulatory inspections.
Sterile Water for Injection (Sterile WFI)
USP References:
- Sterile Water for Injection monograph
- USP <1231> Water for Pharmaceutical Purposes
Sterile Water for Injection is derived from Water for Injection, then sterilized and packaged as a finished pharmaceutical product. Once packaged, it is no longer managed as a water utility. Unlike Purified Water and Water for Injection systems, which are generated, stored, and distributed on site as continuous utilities, Sterile WFI is controlled through batch production, sterility assurance, and quality release and is not distributed through a piping system.
Key distinctions:
- Produced from WFI
- Sterilized and packaged
- Intended for dilution or reconstitution of sterile products
- Not intended for cleaning, rinsing, or bulk distribution
Confusing WFI with Sterile WFI represents a fundamental misunderstanding of USP requirements and is frequently cited during inspections.
Intended Use as the Governing Principle
USP <1231> establishes intended use as the primary determinant of water quality acceptability. Intended use governs:
- Selection of the USP water category
- Requirement for endotoxin control
- Acceptable system design features
- Monitoring parameters and testing frequency
- Qualification scope and depth
Any expansion of use beyond the original justification requires formal evaluation and change control.
Common Industry Missteps
Observed repeatedly during inspections:
- Treating WFI and Sterile WFI as interchangeable
- Using Purified Water for borderline sterile applications
- Applying USP specifications without use-based rationale
- Expanding use without documented impact assessment
Inspectors expect a clear, USP-referenced answer to a simple question: Why is this water acceptable for this use?
Summary
Pharmaceutical water quality categories exist to enforce use-driven, risk-appropriate control. When intended use is clearly defined and aligned with USP requirements, compliance decisions become straightforward, defensible, and sustainable over time.
