Utility Systems in GMP Manufacturing
Utility systems are integral to pharmaceutical manufacturing operations and play a critical role in maintaining controlled conditions, supporting process execution, and protecting product quality. These systems are not ancillary infrastructure. When improperly designed, qualified, or controlled, utility systems are a frequent source of regulatory observations and product quality risk.
Direct and Indirect Impact Utility Systems
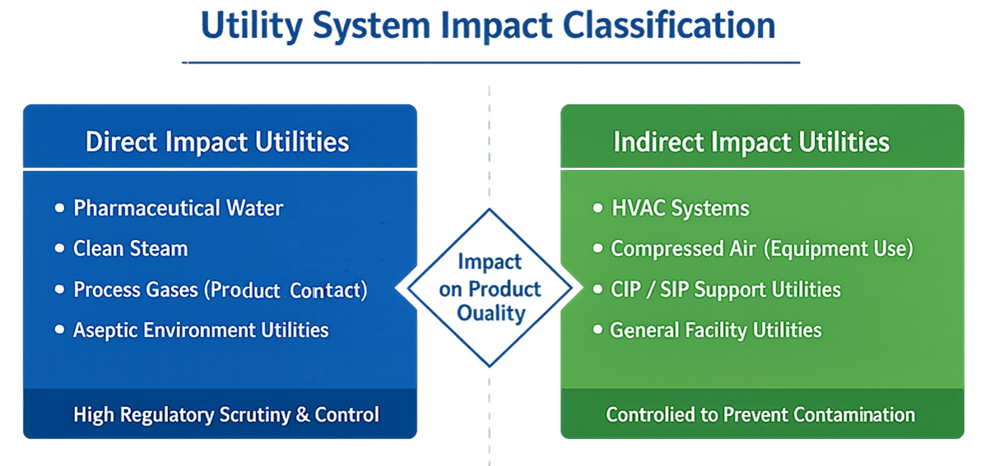
In GMP manufacturing, utility systems are classified based on their potential impact on product quality.
Direct impact utilities are systems that contact product, product contact surfaces, or critical process environments. Examples include pharmaceutical water, clean steam, process gases used in product contact applications, and utilities supporting aseptic processing environments. These systems are subject to the highest level of regulatory scrutiny and require comprehensive qualification, monitoring, and control.
Indirect impact utilities do not contact product directly but support manufacturing operations in a way that could affect product quality if not properly controlled. Examples include HVAC systems serving controlled areas, compressed air used for equipment operation, and utilities supporting cleaning and sterilization processes. Indirect impact utilities must be designed and controlled to prevent contamination, cross-contamination, and operational failures.
This classification drives qualification scope, monitoring requirements, and requalification strategy.
Regulatory Expectations for Utility Systems
Regulatory requirements for utility systems are established through FDA current Good Manufacturing Practice regulations, applicable USP chapters, and recognized industry standards. While regulations do not prescribe specific system designs, they require that utility systems be fit for their intended use and capable of consistently supporting compliant manufacturing operations.
Key regulatory expectations include documented design intent, appropriate qualification and verification, defined operating and quality limits, routine monitoring, effective change control, and periodic evaluation of system performance. Failure to maintain control of utility systems is commonly cited during regulatory inspections and may result in product quality impact or compliance action.
Utility System Lifecycle Perspective
Utility systems are managed using a lifecycle approach that begins with design and risk assessment and continues through installation, qualification, routine operation, monitoring, change control, and requalification. This lifecycle perspective ensures that system performance remains consistent over time and that changes are evaluated based on risk rather than convenience.
This article establishes the foundational principles applicable to all GMP utility systems. Subsequent articles address specific utility types and their unique design, qualification, and monitoring considerations within this common lifecycle framework.
