Validation Steps and the V-Model
Validation of facilities, utilities, equipment, processes, and computerized systems is a foundational activity in pharmaceutical manufacturing and medical device production. Its purpose is to provide documented evidence that systems are designed, installed, operated, and maintained in a manner that consistently meets predefined requirements, regulatory expectations, and intended use.
Validation is not a single activity but a structured lifecycle. The V-model, also referred to as the Verification and Validation model, provides a systematic framework that links design and specification activities on the left side of the model with corresponding verification and qualification activities on the right. This approach is widely applied across regulated industries because it ensures traceability, consistency, and objective evidence throughout the system lifecycle.
Overview of the V-Model
The V-model is represented graphically as the letter “V”:
- The left side represents definition and design stages, where requirements and specifications are developed.
- The right side represents verification and qualification stages, where those requirements are tested and confirmed.
Each design or specification stage has a corresponding qualification or verification activity. This structure enforces traceability from user requirements through final system performance.
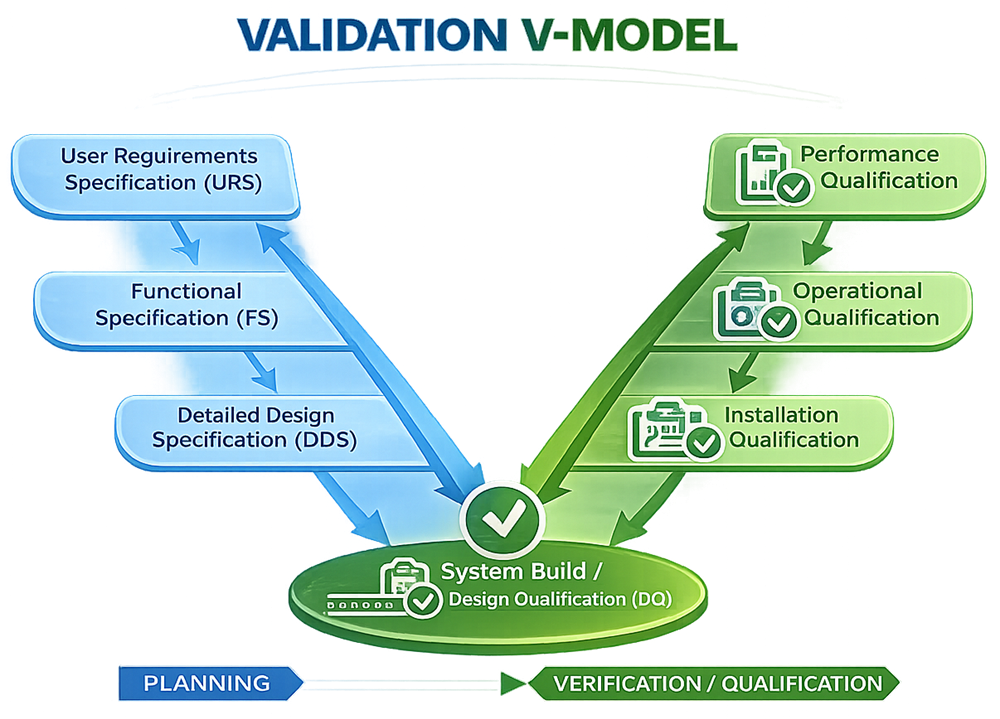
User Requirements Specification (URS)
The User Requirements Specification (URS) defines what the system must do from the user and business perspective. It captures high-level functional, operational, regulatory, and performance requirements without prescribing how the system will be implemented.
The URS establishes the foundation for all subsequent design, procurement, and validation activities. It serves as the primary reference for assessing design adequacy and for demonstrating that the final system is fit for its intended use.
Functional Design Specification (FDS)
The Functional Design Specification (FDS) describes how the system will meet the requirements defined in the URS. It translates user needs into functional descriptions, system behaviors, and interactions.
The FDS focuses on system functionality, logic, and major interfaces, providing a structured description of how the system is expected to operate without yet detailing implementation-level specifics.
Detailed Design Specification (DDS)
The Detailed Design Specification (DDS) expands upon the functional design and defines how the system will be implemented in practice. This includes detailed engineering drawings, configuration parameters, control logic, hardware layouts, software design details, and component-level specifications.
The DDS serves as the technical blueprint for system construction, configuration, and installation and must remain aligned with both the FDS and URS.
Design Qualification (DQ)
Design Qualification (DQ) verifies and documents that the proposed design is suitable to meet the requirements defined in the URS. DQ is typically performed through documented reviews of the URS, FDS, DDS, and supporting design documentation.
The objective of DQ is to confirm that the design is complete, accurate, and appropriate before system installation or implementation begins. DQ establishes confidence that downstream qualification activities are based on an acceptable and compliant design.
Installation Qualification (IQ)
Installation Qualification (IQ) confirms that the system is installed correctly and in accordance with approved design documentation, manufacturer recommendations, and applicable standards.
IQ activities typically include verification of physical installation, configuration, utilities, environmental conditions, software installation, and documentation completeness. Supporting evidence such as as-built drawings, manuals, certificates, and calibration records is reviewed and documented during this phase.
Operational Qualification (OQ)
Operational Qualification (OQ) verifies that the system operates as intended across its defined operating ranges. Testing focuses on functional operation, controls, alarms, interlocks, and system responses under normal and anticipated conditions.
Acceptance criteria are predefined, and test results are documented to demonstrate that the system consistently meets functional and regulatory expectations. OQ establishes that the system performs correctly prior to routine use.
Performance Qualification (PQ)
Performance Qualification (PQ) demonstrates that the system performs effectively and consistently under actual or simulated routine operating conditions. PQ confirms that the system supports real-world use and meets user needs over time.
PQ testing may involve extended operation, representative loads, production simulations, or process runs, depending on system criticality and risk. Successful PQ provides objective evidence that the system is fit for ongoing operational use.
Summary
The V-model provides a structured, traceable approach to validation by linking system requirements and design activities with corresponding qualification and verification phases. By enforcing alignment between specifications and testing, the V-model supports regulatory compliance, risk control, and long-term maintenance of the validated state.
When applied correctly, the V-model ensures that validation activities are planned, executed, and documented in a disciplined and defensible manner across the entire system lifecycle.
