Validation vs Qualification
Validation and qualification are closely related but distinct concepts used in regulated pharmaceutical, biotechnology, and medical device environments. Both activities aim to establish documented evidence of control and compliance, but they differ in scope, intent, and application.
Understanding the distinction between validation and qualification is essential for defining appropriate validation strategies and maintaining regulatory compliance.
Validation
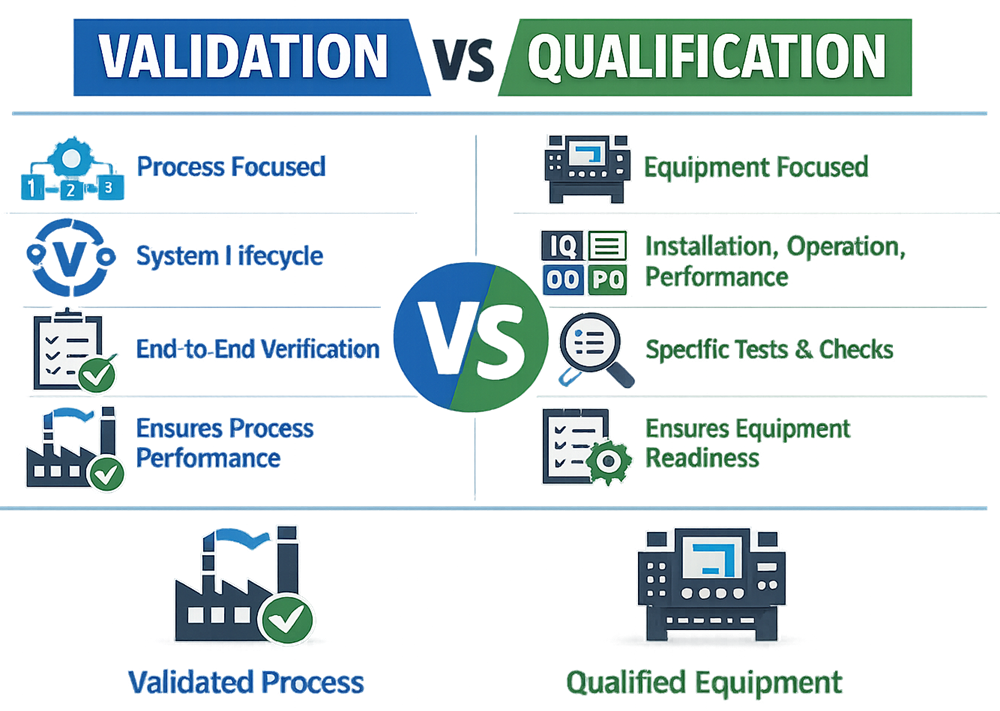
Validation is a broader lifecycle concept that encompasses the activities performed to demonstrate that a process, system, or method consistently produces results that meet predefined requirements, quality attributes, and regulatory expectations.
Validation focuses on fitness for intended use, long-term reliability, and sustained control. It applies to activities that directly impact product quality, patient safety, data integrity, or regulatory compliance.
Key characteristics of validation include:
- Lifecycle-based planning and execution
- Risk-based decision making
- Demonstration of consistent performance over time
- Integration of design, qualification, operation, and monitoring
- Ongoing maintenance of the validated state
Validation does not end with initial testing. It continues throughout the operational lifecycle through change control, monitoring, requalification, and periodic review.
Qualification
Qualification is a subset of validation focused on establishing documented evidence that equipment, systems, utilities, or facilities are designed, installed, and operated in accordance with predefined specifications and requirements.
Qualification activities confirm that a system is suitable to support its intended use and provide the foundation upon which validated processes can operate.
Qualification typically applies to:
- Equipment
- Utilities
- Facilities and controlled environments
- Automated and computerized systems
Qualification is generally executed through structured phases, such as design, installation, operational, and performance qualification, depending on system complexity and criticality.
Relationship Between Validation and Qualification
Validation and qualification are not interchangeable terms.
- Validation addresses whether a process or system consistently delivers the intended outcome.
- Qualification addresses whether the supporting systems are properly designed, installed, and operated to enable that outcome.
Qualification activities support validation, but qualification alone does not demonstrate sustained process performance or control. Validation encompasses qualification and extends beyond it.
Regulatory Perspective
Regulatory agencies expect both validation and qualification to be applied appropriately and proportionally based on risk and intended use. Validation strategies should clearly define which systems require qualification, how qualification supports validated processes, and how the validated state is maintained over time.
Clear distinction and correct application of these concepts reduce compliance risk and improve inspection readiness.
Summary
Validation and qualification are complementary activities with different scopes and objectives. Qualification establishes that systems are suitable and correctly implemented. Validation demonstrates that processes and systems consistently achieve their intended purpose. Together, they form a structured and defensible approach to compliance and quality assurance in regulated environments.
