VHP Sterilization Process Overview
1. Introduction
Vapor Hydrogen Peroxide VHP sterilization is a widely adopted low-temperature sterilization technology used in pharmaceutical and sterile manufacturing environments. It is primarily applied for the decontamination and sterilization of isolators, RABS, material transfer chambers, pass-through units, and occasionally cleanroom spaces.
VHP sterilization in regulated environments, it is an engineered, controlled, and repeatable process integrated into facility and equipment design. Its performance depends as much on system architecture and airflow control as on chemical lethality. When properly designed and operated, VHP systems can consistently achieve a sterility assurance level appropriate for aseptic processing support applications.
2. Fundamental Principles of VHP Sterilization
Hydrogen peroxide vapor is a strong oxidizing agent. Microbial inactivation occurs through oxidative damage to:
- Cell membranes
- Proteins and enzymes
- DNA and nucleic acids
The lethality of VHP depends on several interacting factors:
- Vapor concentration
- Exposure time
- Temperature
- Relative humidity
- Surface accessibility
Unlike steam sterilization, VHP sterilization is a surface process. Effective sterilization requires direct vapor contact with exposed surfaces. Any area shielded from vapor flow, excessively absorbent materials, or geometrically restricted locations may compromise performance.
VHP is most effective under controlled humidity conditions. Excess moisture can lead to condensation and micro-condensation phenomena, while insufficient humidity may reduce sporicidal efficiency. Therefore, humidity control is not incidental; it is integral to process performance.
3. Typical VHP Cycle Phases
Although specific cycle designs vary by manufacturer and application, VHP sterilization cycles typically consist of conditioning, injection, dwell, and aeration phases, each controlled to achieve predictable and repeatable decontamination performance.
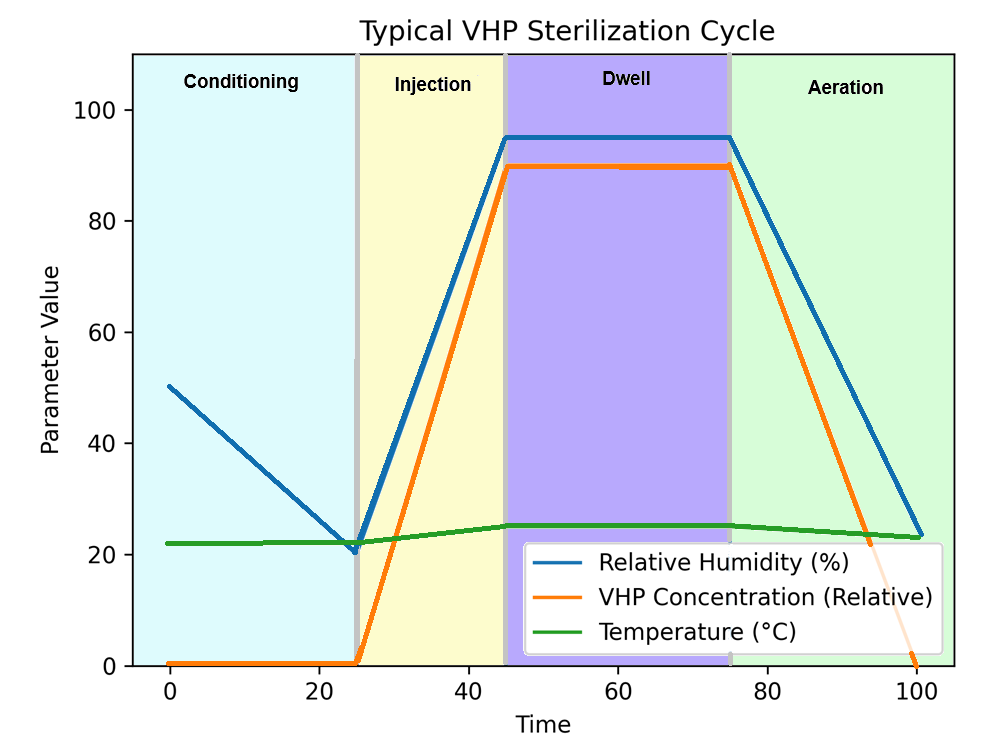
3.1 Conditioning or Dehumidification
The enclosure is first conditioned to reduce relative humidity to a defined setpoint. Objectives of this phase:
- Remove excess moisture
- Establish consistent starting conditions
- Improve vaporization efficiency
- Reduce risk of condensation
Failure to properly condition the chamber may result in unpredictable distribution or localized condensation.
3.2 VHP Injection
Liquid hydrogen peroxide solution is vaporized and introduced into the enclosure. Key elements include:
- Controlled vaporization rate
- Uniform distribution through circulation systems
- Continuous monitoring of injection parameters
At this stage, concentration rises toward a target value. Airflow design and internal geometry significantly influence distribution uniformity.
3.3 Dwell or Exposure Phase
During dwell, target concentration is maintained for a defined period. This phase determines lethality and must ensure:
- Adequate concentration stability
- Uniform vapor distribution
- Absence of untreated shadowed areas
Surface temperature and material composition influence vapor interaction and potential absorption. Materials with high peroxide absorption capacity may act as sinks, reducing available concentration.
3.4 Aeration
Following exposure, the system transitions to aeration. Objectives:
- Catalytic decomposition of hydrogen peroxide into water and oxygen
- Removal of residual vapor
- Reduction of concentration to safe re-entry levels
Aeration performance depends on:
- Catalyst efficiency
- Air exchange rate
- Chamber tightness
- Load characteristics
Inadequate aeration may result in excessive residual peroxide levels, posing safety and product compatibility concerns.
4. VHP Equipment Design and System Architecture
Process performance cannot be separated from equipment design. In regulated applications, VHP sterilization must be treated as an engineered system rather than a portable fumigation device.
Example of a large-scale industrial VHP sterilization chamber with integrated external generation skid used in pharmaceutical manufacturing environments.

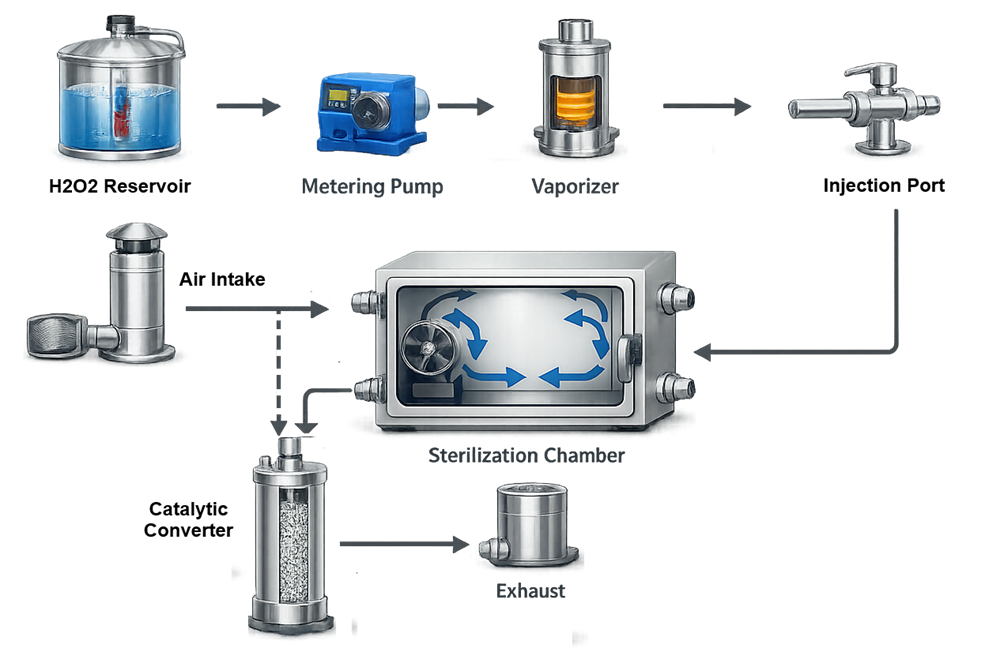
4.1 Core System Components
A typical VHP system includes:
- Hydrogen peroxide storage reservoir
- Metering pump or injection system
- Vaporizer or flash evaporator
- Distribution manifold
- Circulation fans
- Catalytic converter for aeration
- Control system with HMI
- Safety interlocks and environmental monitoring
Material compatibility is critical. Stainless steel grades, elastomers, gaskets, gloves, and filters must be compatible with repeated peroxide exposure. Dead legs, condensate traps, and stagnant zones should be eliminated during design. Internal geometry must support predictable vapor circulation.
4.2 Chamber and Isolator Design Considerations
The enclosure being sterilized is part of the sterilization system. Design attributes influencing performance include:
- Leak-tight construction
- Surface finish and weld quality
- Internal layout and obstructions
- Glove ports and transfer interfaces
- Pass-through integration
- Door seals and gasket integrity
Airflow patterns during injection and aeration must be understood and characterized. Poor airflow design results in stratification and shadowing. VHP sterilization effectiveness is inseparable from enclosure architecture.
Illustration below showing internal airflow patterns within a VHP sterilization chamber and the formation of reduced-exposure regions behind load obstructions.

4.3 Airflow and Distribution Control
Uniform vapor distribution requires:
- Engineered circulation paths
- Defined inlet and return locations
- Adequate fan capacity
- Balanced flow dynamics
Complex internal geometries, shelving, or equipment loads create areas of restricted access. These areas typically define worst-case locations for subsequent validation.
4.4 Monitoring and Control Systems
A properly designed VHP system includes monitoring of:
- Temperature
- Relative humidity
- Pressure
- Injection rate
- Cycle timing
- System status and alarms
Some systems include direct hydrogen peroxide concentration monitoring. Others rely on controlled injection algorithms. The control system must ensure:
- Repeatability
- Traceable cycle records
- Alarm management
- Operator safety
Safety features typically include door interlocks, peroxide leak detection, and emergency abort functions.
5. Process Variables and Critical Parameters
A distinction must be made between process variables and critical parameters.
Process Variables
These influence the process but may not be independently controlled at a fixed setpoint. Examples:
- Load configuration
- Surface materials
- Chamber geometry
Critical Parameters
These must be controlled within defined limits to ensure performance. Typically include:
- VHP concentration or injection rate
- Exposure time
- Temperature
- Relative humidity
- Airflow rate
Understanding which parameters are critical requires risk-based evaluation and empirical confirmation during validation.
6. Typical Failure Modes and Operational Risks
In practice, VHP sterilization failures most often arise from engineering or operational weaknesses rather than chemistry. Common issues include:
- Inadequate dehumidification leading to condensation
- Poor vapor distribution due to airflow imbalance
- Excessive absorption by packaging or porous materials
- Chamber leakage reducing effective concentration
- Catalyst degradation impairing aeration
- Inconsistent load configuration
A mature VHP program anticipates these risks and designs controls accordingly.
7. Operational Considerations
Routine operation requires:
- Defined load configuration
- Standardized cycle parameters
- Operator training
- Preventive maintenance of injection and catalytic systems
- Periodic leak integrity testing
Operational discipline is essential. VHP sterilization is not forgiving of uncontrolled variability.
8. Engineering Control and Operational Reliability
In regulated pharmaceutical environments, VHP sterilization must operate as a controlled and repeatable engineered process. Performance consistency depends on the integrity of system design, operational discipline, and preventive maintenance. Key elements of sustained reliability include:
- Stable and repeatable cycle parameter control
- Verified airflow performance and distribution integrity
- Routine inspection of seals, gloves, and enclosure tightness
- Proper maintenance of vaporization and catalytic aeration systems
- Defined and standardized load configurations
- Monitoring of environmental and safety interlocks
Degradation in mechanical components, airflow systems, or enclosure integrity can materially affect process performance. Therefore, the VHP system must be treated as critical process equipment and managed accordingly. Operational controls must ensure that:
- Cycle parameters remain within defined limits
- Equipment components remain in a qualified mechanical state
- Safety systems function as intended
- Operator practices do not introduce uncontrolled variability
When the engineering design is sound and operational controls are disciplined, VHP sterilization provides a reliable and predictable decontamination solution suitable for aseptic manufacturing support applications.qualification strategy, biological indicator placement, acceptance criteria, requalification triggers, and continued verification.
