Pharmaceutical Water System Lifecycle Control and Requalification
Pharmaceutical water systems require ongoing lifecycle control to maintain a sustained state of compliance. Initial qualification establishes baseline capability, but continued control is demonstrated through structured monitoring, change control, periodic review, and justified requalification activities.
Lifecycle control ensures that water quality remains aligned with intended use as systems age, usage evolves, and operating conditions change.
Lifecycle Control Principles
Lifecycle control of pharmaceutical water systems is based on four core principles:
- Water quality must remain aligned with intended use
- System performance must be continuously monitored and evaluated
- Changes must be assessed for impact before implementation
- Requalification must be risk-based and justified
These principles are consistent with expectations described in USP <1231> and routinely evaluated during FDA inspections.
Routine Monitoring as the Primary Control Mechanism
Routine monitoring is the primary mechanism for demonstrating continued control of pharmaceutical water systems. Monitoring programs typically include:
- Online monitoring of critical parameters such as conductivity, temperature, and flow
- Periodic offline testing for microbiological quality and endotoxins where applicable
- Defined alert and action limits
- Formal trending and periodic review
Monitoring data provide early indication of performance drift and support timely intervention before specifications are exceeded.
Periodic Review Checklist for Pharmaceutical Water Systems
Periodic review is performed to confirm that the pharmaceutical water system remains fit for its intended use and continues to operate in a sustained state of control. The following elements should be evaluated and documented during each periodic review.
1. System Description and Scope Confirmation
- Water quality category and intended use remain unchanged
- System configuration reflects the current as-built condition
- Points of use remain appropriate for system capacity and design
2. Monitoring and Trending Review
- Review of conductivity, TOC, microbiological, and endotoxin trends as applicable
- Assessment of long-term stability and variability
- Evaluation of recurring alert-level results
- Confirmation that action limits have not been exceeded without investigation
3. Sanitization Program Review
- Sanitization frequency remains appropriate for system performance
- Sanitization methods executed as defined
- Evidence of sanitization effectiveness reviewed
- Any sanitization failures or deviations evaluated
4. Sampling Program Adequacy
- Sampling locations remain representative of system risk
- Worst-case and remote points of use remain appropriate
- Sampling frequency justified based on performance history
- No unexplained gaps in sampling coverage
5. Deviations, Alerts, and Investigations
- Summary of deviations related to water system performance
- Review of investigations and root cause conclusions
- Assessment of effectiveness of corrective and preventive actions
- Evaluation of recurrence or emerging patterns
6. Maintenance and Operational Changes
- Review of maintenance activities since last review
- Assessment of maintenance impact on system performance
- Verification that post-maintenance checks were performed
- Identification of any operational changes affecting system use
7. Change Control Review
- Review of approved changes affecting the water system
- Confirmation that impact assessments were performed
- Verification that required testing or qualification activities were completed
- Assessment of cumulative impact of multiple changes
8. Requalification Assessment
- Evaluation of defined requalification triggers
- Determination of whether requalification is required
- Justification of requalification scope or rationale for no action
- Documentation of decisions and approvals
9. Regulatory and Standard Updates
- Review of relevant updates to USP requirements
- Assessment of impact of regulatory guidance changes
- Confirmation that system controls remain aligned with expectations
10. Review Conclusion
- Overall assessment of system state of control
- Confirmation that water quality remains fit for intended use
- Identification of required follow-up actions
- Approval and documented conclusion of periodic review
Periodic Review Conclusion
Periodic review provides documented confirmation that the pharmaceutical water system remains in a state of control and continues to support its intended use. Review outcomes are used to determine the need for corrective actions, changes to monitoring, or requalification activities.
Change Control and Impact Assessment
All changes to pharmaceutical water systems must be managed through formal change control. Changes requiring evaluation include:
- Equipment replacement or modification
- Changes to operating parameters or control strategies
- Sanitization method changes
- Addition, removal, or relocation of points of use
- Changes in water demand or intended use
Each change must be evaluated for potential impact on:
- Chemical quality
- Microbiological control
- Endotoxin control
- Monitoring effectiveness
Risk assessment determines whether requalification is required and defines its scope.
Requalification Triggers
Requalification is not performed on a fixed schedule by default. It is triggered by evidence or risk, not by time alone.
Common requalification triggers include:
- Repeated alert or action level excursions
- Adverse performance trends
- Sanitization failures or loss of effectiveness
- Significant system modifications
- Extended shutdowns or prolonged idle conditions
- Changes in regulatory expectations or intended use
Triggers must be clearly defined and consistently applied. The following diagram illustrates a structured, risk-based approach for determining requalification scope for pharmaceutical water systems based on identified triggers, impact assessment, and risk evaluation.
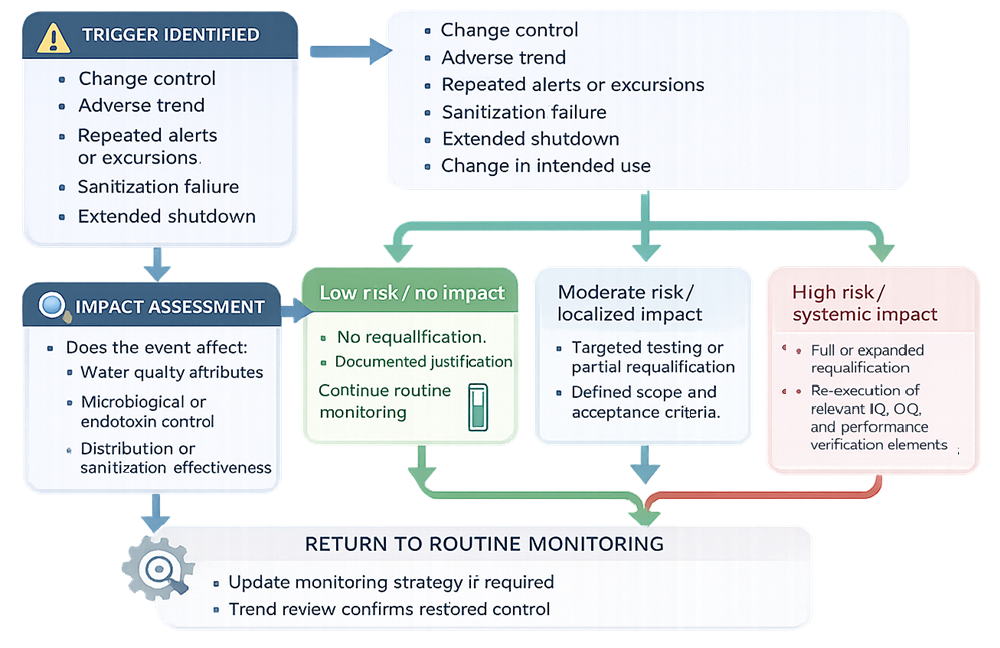
Determination of Requalification Scope
Requalification scope is determined based on:
- Nature and extent of the change or issue
- Risk to product quality
- Historical system performance
- Effectiveness of existing controls
Requalification may range from:
- Targeted testing or document review
- Partial re-execution of qualification activities
- Full requalification of the system
The objective is to restore confidence in system control without unnecessary testing.
Integration with Deviation Management
Water system performance issues must be integrated with deviation and investigation processes. Investigations should:
- Identify root cause rather than symptoms
- Evaluate impact on product and operations
- Assess whether existing controls remain effective
- Determine whether requalification is required
Failure to link deviations with lifecycle control is frequently cited during inspections.
Documentation and Governance
Lifecycle control activities must be supported by controlled documentation, including:
- Monitoring and trending records
- Periodic review reports
- Change control documentation
- Requalification justifications and reports
Documentation must demonstrate active system oversight, not passive record keeping.
Common Lifecycle Control Deficiencies
Frequently observed deficiencies include:
- Monitoring programs that detect issues but do not drive action
- Inadequate trending and periodic review
- Changes implemented without appropriate impact assessment
- Requalification performed inconsistently or without justification
- Overreliance on initial qualification data
These gaps undermine confidence in continued system control.
Summary
Pharmaceutical water system lifecycle control ensures that initial qualification remains meaningful throughout the operational life of the system. Effective lifecycle control relies on disciplined monitoring, structured change control, periodic performance review, and risk-based requalification.
When lifecycle control is properly implemented, water systems remain compliant, reliable, and aligned with intended use despite change and aging.
