Pharmaceutical Water System Qualification and Performance Verification
Pharmaceutical water systems are direct-impact GMP utilities and must be qualified using a structured, risk-based approach. Qualification activities must demonstrate that the system, as installed and operated, can reliably produce water meeting defined quality requirements at all points of use and under all expected operating conditions.
Qualification scope and depth are determined by intended use, water quality category, system complexity, and risk to product quality.
Qualification Strategy and Risk Assessment
Water system qualification begins with a documented risk assessment that defines:
- Critical quality attributes applicable to the water type
- System elements that directly affect chemical, microbiological, and endotoxin control
- Sampling locations and worst-case conditions
- Qualification scope and acceptance criteria
Risk assessment drives:
- IQ and OQ test coverage
- Performance verification sampling plans
- Sanitization verification requirements
- Requalification triggers
This approach aligns with expectations described in USP <1231> and routinely evaluated by FDA during inspections. The following diagram illustrates the lifecycle approach used for qualification and performance verification of pharmaceutical water systems
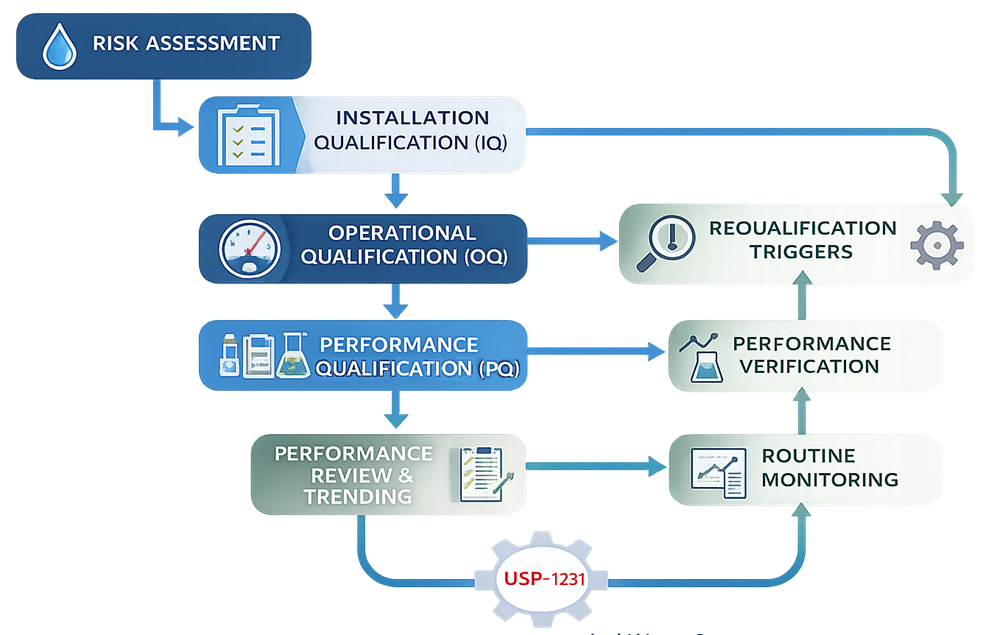
Installation Qualification (IQ)
Installation Qualification verifies that the water system is installed in accordance with approved design documentation and manufacturer recommendations. IQ activities typically include:
- Verification of equipment installation against drawings and specifications
- Confirmation of materials of construction
- Verification of piping slope, dead-leg control, and system configuration
- Documentation of weld quality and surface finish where applicable
- Verification of installed instrumentation and utilities
IQ establishes the baseline physical configuration of the system.
Operational Qualification (OQ)
Operational Qualification verifies that system controls, alarms, and operational functions perform as intended across defined operating ranges. OQ typically includes:
- Control system functionality and interlocks
- Alarm setpoints and response verification
- Flow, pressure, temperature, and circulation verification
- Execution of sanitization sequences
- Startup, shutdown, and recovery testing
OQ focuses on functional performance, not long-term water quality results.
Verification of Sanitization Methods
Sanitization is a critical control mechanism and must be explicitly verified as part of qualification. Sanitization verification includes:
- Confirmation that sanitization methods reach all intended system elements
- Verification of critical sanitization parameters such as temperature, concentration, or contact time
- Demonstration that sanitization effectively reduces microbial load
- Documentation of sanitization execution and acceptance criteria
Both routine and worst-case sanitization scenarios must be considered based on system design and risk.
Performance Qualification and Performance Verification
Performance Qualification or performance verification demonstrates that the water system can consistently meet USP quality requirements during routine operation. This phase focuses on:
- Chemical quality
- Microbiological control
- Endotoxin control where applicable
- System stability over time
Sampling plans must be risk-based and justified, not arbitrarily extensive.
Consideration of Seasonal and Operating Variability
Performance verification must account for normal variability, including:
- Seasonal changes in feed water quality
- Variations in ambient temperature and humidity
- Changes in demand and system usage
- Effects of routine maintenance and sanitization
Where seasonal variation is expected, performance verification must include sufficient duration or justification to demonstrate system robustness.
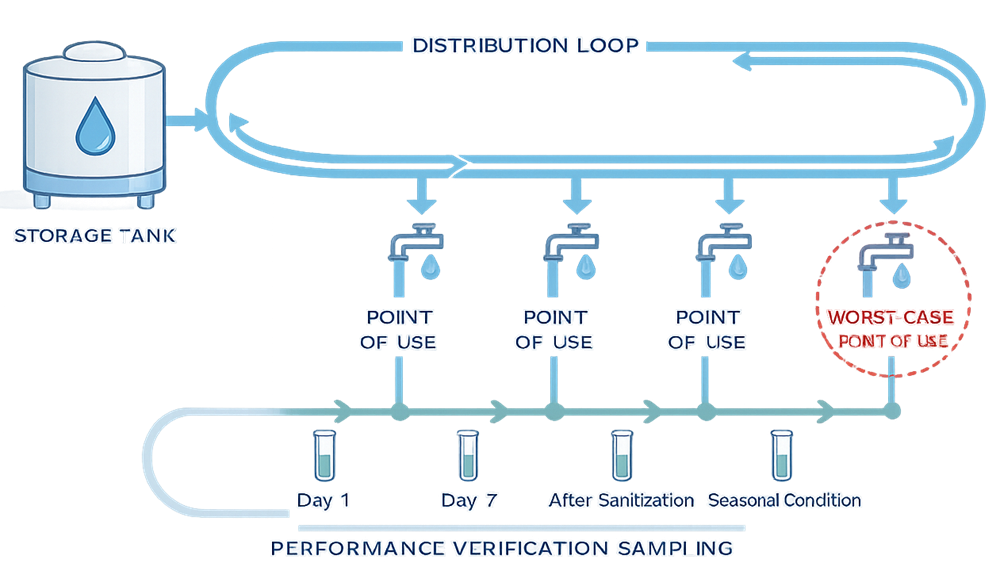
Sampling Strategy During Performance Verification
Performance verification sampling programs typically include:
- Multiple points of use
- Worst-case and most remote locations
- Sampling before and after sanitization
- Sampling over a defined time period sufficient to demonstrate stability
The objective is to confirm consistent performance, not to generate excessive data.
Acceptance Criteria and Data Evaluation
Acceptance criteria are based on:
- Applicable USP monographs
- Internal specifications aligned with intended use
- Alert and action limits defined in monitoring programs
Results must be evaluated collectively, with emphasis on:
- Trends
- Variability
- Repeated alert-level results
Isolated passing results do not demonstrate control.
Qualification Scope by Pharmaceutical Water Type
The scope and depth of qualification activities vary by pharmaceutical water category and intended use. Higher-risk water types require broader verification of controls, sanitization, and performance.
| Qualification Element | Purified Water (PW) | Water for Injection (WFI) | Sterile Water for Injection |
|---|---|---|---|
| Installation Qualification (IQ) | Required | Required | Not applicable to utility system |
| Operational Qualification (OQ) | Required | Required | Not applicable to utility system |
| Performance Qualification / Verification | Required | Required | Not applicable to utility system |
| Chemical Quality Verification | Required | Required | Verified as finished product |
| Microbiological Performance Verification | Required | Required with enhanced scrutiny | Sterility testing required |
| Endotoxin Verification | Not required unless justified by use | Required | Required |
| Sanitization Method Verification | Required | Required with defined effectiveness criteria | Not applicable to utility system |
| Seasonal / Feed Water Variability Consideration | Required | Required | Not applicable |
| Worst-Case Point-of-Use Sampling | Required | Required | Not applicable |
| Transition to Routine Monitoring | Required | Required | Not applicable |
| Lifecycle Monitoring and Trending | Required | Required | Managed as finished product |
| Requalification Triggers Defined | Required | Required | Managed under product lifecycle |
Notes for Clarity and Inspection Readiness
- Sterile Water for Injection is excluded from utility qualification activities because it is a finished, packaged pharmaceutical product, not a distributed water system.
- Endotoxin verification is the key differentiator between PW and WFI qualification scope.
- Sanitization verification is expected for all distributed utility systems, regardless of water type.
- Seasonal variability applies only to on-site generated utilities, not packaged products.
Transition to Routine Monitoring
Successful completion of performance verification supports transition to routine monitoring.
This transition includes:
- Establishment of routine sampling locations and frequencies
- Implementation of online and offline monitoring programs
- Formal trending and periodic review
- Integration with deviation management and change control
Qualification data provide the baseline against which ongoing performance is evaluated.
Requalification Considerations
Requalification is not performed on a fixed schedule by default. It is triggered by:
- Changes to system configuration or use
- Repeated deviations or adverse trends
- Sanitization failures
- Extended shutdowns or major maintenance
- Changes in regulatory expectations
Requalification scope must be justified based on risk and historical performance.
Common Qualification Weaknesses
Frequently observed deficiencies include:
- Qualification performed without documented risk assessment
- Sanitization methods assumed but not verified
- Inadequate consideration of seasonal variability
- Poor linkage between qualification data and routine monitoring
- Failure to define requalification triggers
These weaknesses are routinely cited during inspections.
Summary
Pharmaceutical water system qualification and performance verification provide documented evidence that the system is capable of sustained, compliant operation. Effective qualification is risk-based, technically grounded, and directly linked to routine monitoring and lifecycle control.
When properly executed, qualification supports long-term system reliability and regulatory confidence.
